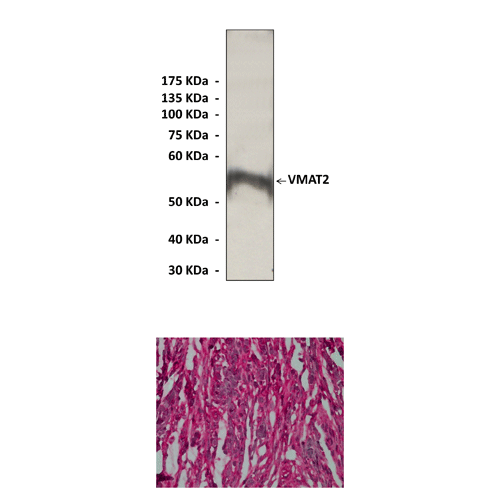
Anti-VMAT2: Rabbit VMAT2 Antibody |
 |
BACKGROUND Vesicular monoamine transporter (VMAT2) is a 12-transmembrane domain H+-ATPase antiporter, which uses an electrochemical gradient to drive transport; two protons are exchanged for one monoamine molecule. Phylogenetically, VMAT2 is a member of the solute carrier protein family and the toxin-extruding antiporter (TEXAN) gene family, which includes bacterial resistance genes. Moreover, VMAT2 contains sequence homology and functional similarities to the major facilitator superfamily of drug resistance transporters. The brain VMAT2 localizes on vesicular membranes in neurons and acts to accumulate monoamine neurotransmitters including dopamine, serotonin, norepinephrine, and histamine from neuronal cytoplasm into synaptic vesicles after they have been synthesized from their precursors for regulated exocytotic release. Normal vesicular monoamine release through calcium-dependent vesicle fusion with presynaptic membranes is thus dependent on normal function of VMAT2.1 VMAT2 has a similar selectivity for all monoamines and is present throughout the central and periphery nervous system. It has been suggested that VMAT2 has evolved to serve an analogous role in eukaryotic systems by providing a mechanism to sequester and clear toxins from the cell. Thus, vesicular sequestration serves a dual purpose: preventing the interaction of toxins with molecular machinery and limiting exposure of neighboring cells to the toxin. In fact, VMAT2 was partly identified via its ability to confer resistance to the dopaminergic toxin 1-methyl-4-phenylpyridinium (MPP+), which is commonly used to induce a Parkinsonian phenotype in mice. The sequestration of monoamines is important for maintenance of normal neurotransmission and also acts to keep intracellular levels of the monoamines below potentially toxic levels. The level of VMAT2 expression is essential to proper monoaminergic handling, as it regulates both the size of the vesicular monoamine pool and influences the availability of monoamines in the cytosol, influencing cellular susceptibility to oxidation. The monoamines, particularly DA and norepinephrine (NE) have the ability to spontaneously oxidize in the cytosol, potentially damaging cellular machinery. VMAT2 has been suggested to be an excellent marker of presynaptic dopaminergic nerve terminals in the striatum of Parkinson's disease patients based on its high level of expression and insensitivity to drugs used to treat the disease. It was demonstrated that reduced vesicular storage of monoamines and the resulting disruption of the cytosolic environment may play a role in the pathogenesis of Parkinsonian symptoms and neurodegeneration.2 The multisystem nature of the VMAT2-deficient mice may be useful in developing therapeutic strategies that go beyond the dopamine system. In addition, except the central and peripheral nervous system, VMAT2 is also expressed in the hematopoietic system including in mast cells and platelets, and the neuroendocrine system. In the neuroendocrine system, VMAT2 is expressed in chromaffin cells of the adrenal medulla, histamine-storing enterochromaffin-like cells of the stomach, and pancreatic islet cells (VMAT2 mRNA is more abundant in isolated islets than exocrine pancreas in humans). Moreover, VMAT2 is constantly expressed in pancreatic beta cells and has recently been proposed as a target for measurement of beta cell mass in vivo.3
REFERENCES
1. Uhl, G.R. et al: FASEB J.14:2459-65, 2000
2. Okamura, N. et al: J. Nucl. Med. 51:223-8, 2010
3. Saisho, Y. et al: J Mol Hist (2008) 39:543–551
2. Okamura, N. et al: J. Nucl. Med. 51:223-8, 2010
3. Saisho, Y. et al: J Mol Hist (2008) 39:543–551
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CY1116 |
Antigen: | A short peptide from human VMAT2 sequence. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 56 kDa |
Specificity/Sensitivity: | Detects endogenous levels of VMAT2 proteins without cross-reactivity with other related proteins. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой