FGF9 ELISA Kit, Human |
 |
BACKGROUND FGFs comprise a large family of proteins that includes at least 22 known members. FGFs bind and signal through low and high affinity FGF receptors. The four known high affinity receptors (FGFR1–4) are structurally similar transmembrane receptor tyrosine kinases. FGF family members possess broad mitogenic and cell survival activities, and are involved in a variety of biological processes, including embryonic development, cell growth, morphogenesis, tissue repair, tumor growth and invasion.1
FGF9 is a 26 kDa secreted protein and is a potent mitogen and survival factor required for morphogenesis during embryonic development and numerous biological functions at adulthood. It plays critical role in epidermal proliferation and wound healing in the skin of mice and has a paracrine role in toxic liver injury. FGF9 acts on its receptors (FGFR2 and 3) to activate intracellular signal transduction cascades on target cells, which affects the expression of downstream target genes including members of the bone morphogenetic protein (BMP) family. The expression of BMP-2, BMP-4 and noggin (the inducible antagonist of BMP-2) are known to affect proliferation and cell survival as a result of FGF9 activation in human tissue. Moreover, FGF9 exhibits a growth-stimulating effect on cultured glial cells. In nervous system, this protein is produced mainly by neurons and may be important for glial cell development. Expression of the mouse homolog of this gene was found to be dependent on Sonic hedgehog (Shh) signaling. Mice lacking the homolog gene displayed a male-to-female sex reversal phenotype, which suggested a role in testicular embryogenesis. Furthermore, FGF9 can regulate multiple SRY (sex-determining region on the Y gene)-dependent processes, such as mesenchymal proliferation and mesonephric migration into the gonad and Sertoli cell differentiation. It was demonstrated that FGF9 produced in the fetal testis acts directly on germ cells to inhibit meiosis; in addition, FGF9 maintains expression of pluripotency-related genes and upregulates markers associated with male germ cell fate.2 It was also reported that FGF9 stimulates steroidogenesis in postnatal Leydig cells. Ras-MAPK, PI3K and PKA signalling pathways are involved in the FGF9-induced steroidogenesis.3 Additionally, FGF9 expression is sufficient to convert the differentiation program of (at least a subset of) mesoderm-derived cranial mesenchyme cells from intramembranous to endochondral ossification.4 Furthermore, FGF9 may be involved in embryonic skeletal development. It play an important role in long bone repair, presumably by initiating angiogenesis through VEGF-A.5
FGF9 is a 26 kDa secreted protein and is a potent mitogen and survival factor required for morphogenesis during embryonic development and numerous biological functions at adulthood. It plays critical role in epidermal proliferation and wound healing in the skin of mice and has a paracrine role in toxic liver injury. FGF9 acts on its receptors (FGFR2 and 3) to activate intracellular signal transduction cascades on target cells, which affects the expression of downstream target genes including members of the bone morphogenetic protein (BMP) family. The expression of BMP-2, BMP-4 and noggin (the inducible antagonist of BMP-2) are known to affect proliferation and cell survival as a result of FGF9 activation in human tissue. Moreover, FGF9 exhibits a growth-stimulating effect on cultured glial cells. In nervous system, this protein is produced mainly by neurons and may be important for glial cell development. Expression of the mouse homolog of this gene was found to be dependent on Sonic hedgehog (Shh) signaling. Mice lacking the homolog gene displayed a male-to-female sex reversal phenotype, which suggested a role in testicular embryogenesis. Furthermore, FGF9 can regulate multiple SRY (sex-determining region on the Y gene)-dependent processes, such as mesenchymal proliferation and mesonephric migration into the gonad and Sertoli cell differentiation. It was demonstrated that FGF9 produced in the fetal testis acts directly on germ cells to inhibit meiosis; in addition, FGF9 maintains expression of pluripotency-related genes and upregulates markers associated with male germ cell fate.2 It was also reported that FGF9 stimulates steroidogenesis in postnatal Leydig cells. Ras-MAPK, PI3K and PKA signalling pathways are involved in the FGF9-induced steroidogenesis.3 Additionally, FGF9 expression is sufficient to convert the differentiation program of (at least a subset of) mesoderm-derived cranial mesenchyme cells from intramembranous to endochondral ossification.4 Furthermore, FGF9 may be involved in embryonic skeletal development. It play an important role in long bone repair, presumably by initiating angiogenesis through VEGF-A.5
REFERENCES
1. Turner, N. & Grose, R.: Nat. Rev. Cancer 10:116-29, 2010
2. Bowles, J. et al: Dev. Cell 19:440-9, 2010
3. Lin, Y.M. et al: Int. J. Androl. 33:545-53, 2010
4. Kim, Y. et al: PLoS Biology 4:e187, 2006
5. Behr, B. et al: Proc. Natl. Acad. Sci. USA 107:11853-8, 2010
2. Bowles, J. et al: Dev. Cell 19:440-9, 2010
3. Lin, Y.M. et al: Int. J. Androl. 33:545-53, 2010
4. Kim, Y. et al: PLoS Biology 4:e187, 2006
5. Behr, B. et al: Proc. Natl. Acad. Sci. USA 107:11853-8, 2010
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CL0348 |
Target Protein Species: | Human |
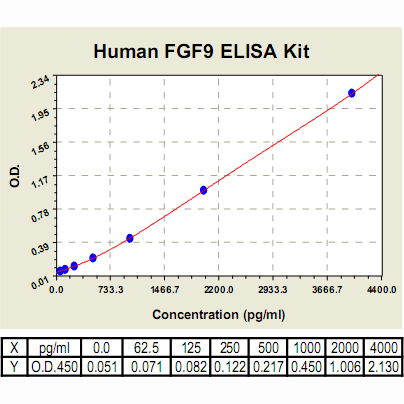
Range: | 62.5 pg/ml – 4000 pg/ml |
Specificity: | No detectable cross-reactivity with other cytokines |
Storage: | Store at 4°C. Use within 6 months. |
ELISA Kits are based on standard sandwich enzyme-linked immunosorbent assay technology. Freshly prepared standards, samples, and solutions are recommended for best results.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой