Anti-Abi-1: Rabbit Abi-1 Antibody |
 |
BACKGROUND The Abl binding protein 1 (Abi1), also known as Hssh3bp1, is a key component of several intrinsic complexes that regulate actin cytoskeletal remodeling near the plasma membrane. Overexpression of Abi1 in NIH 3T3 cells inhibits macropinocytosis. Numerous structurally distinct isoforms of Abi1 exist in mammalian cells providing a potential diversity of Abi1-actin regulatory complexes, suggesting the possible existence of multiple mechanisms through which Abi1 might regulate macropinocytosis.1 Abi1 participates in several multi-protein complexes that regulate the dynamics of actin polymerization. Eps8-Sos1, NWasp, and Wave complex including both Wave 1 and Wave 2, have been proposed to form complexes with Abi1. Each of these actin regulatory complexes involves a different mechanism, although at least two of these mechanisms converge directly on Rac1 function. The first, Eps8-Sos1-Abi1 complex, exhibits Rac-specific guanine exchange factor (GEF) activity transducing signals from Ras to Rac and activating the latter. The second, Abi1-containing Wave 2 complex, has been directly linked to the effect of Rac1 on increased actin polymerization at the plasma membrane at the site of control of lamellipodia formation.2 Mechanistically, it was hypothesized that activated Rac1 targets the Wave 2 complex to the plasma membrane where Rac1 is responsible for activation of Wave 2/Arp2/3-dependent actin polymerization. Increased actin polymerization near the plasma membrane, which is associated with increased macropinocytic uptake, may explain the postulated role of Rac1 in regulation of macropinocytosis. In this regard it has been demonstrated that a dominant negative Rac1 (Rac1-T17N) downregulates fluid uptake, but constitutively active Rac1 (Rac1-Q61L) upregulates the process. In the Wave 2 complex, Rac1 does not interact directly with Wave 2, but binds to Rac1-binding proteins Sra-1 and Nap1, which form complexes with Abi1. Abi1 interacts with Wave 2 and couples Wave 2 complex to Abl kinase activity after cell stimulation, thus promoting Wave 2 phosphorylation. Tyrosine phosphorylation may regulate Wave complex activity either by regulating conformation of the complex or by regulating interactions among components of the complex such as Abi1 and Abl kinase or Abi1 and the p85 regulatory subunit of PI-3 kinase.3
REFERENCES
1. Innocenti, M. et al: Nat Cell Biol 6:319–27, 2004
2. Leng, Y. et al: proc. Natl. Acad. Sci. USA 102: 1098–103, 2005
3. Proepper, C. et al: EMBO J. 26:1397-409, 2007
2. Leng, Y. et al: proc. Natl. Acad. Sci. USA 102: 1098–103, 2005
3. Proepper, C. et al: EMBO J. 26:1397-409, 2007
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CA1001 |
Antigen: | Short peptide from human Abi-1 sequence. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:50 - 1:200 ICC n/d FACS n/d |
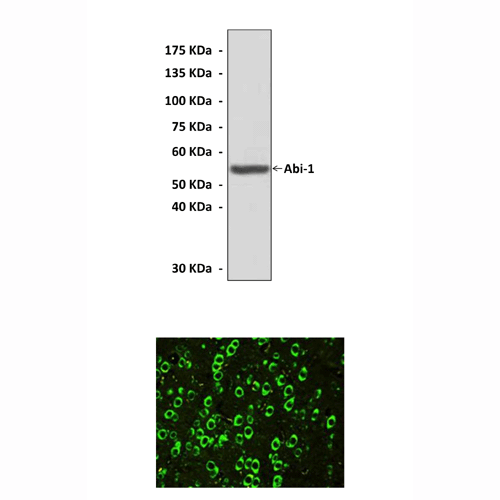
Predicted Molecular Weight of protein: | 56 kDa |
Specificity/Sensitivity: | Detects endogenous levels of Abi-1 proteins without cross-reactivity with other related proteins. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой