Anti-EGF: Mouse Epidermal Growth Factor Antibody |
 |
BACKGROUND EGF is the founding member of the EGF-family of protein. Members of this protein family have highly similar structural and functional characteristics. Human EGF is a 6045-Da protein with 53 amino acid residues and three intramolecular disulfide bonds. Besides EGF itself other family members include: HB-EGF, TGF-alpha, Amphiregulin, Epiregulin, Epigen, Betacellulin, NRG-1, -2, -3, and -4. EGF has a profound effect on the differentiation of specific cells in vivo and is a potent mitogenic factor for a variety of cultured cells of both ectodermal and mesodermal origin. The EGF precursor is believed to exist as a membrane-bound molecule which is proteolytically cleaved to generate the 53-amino acid peptide hormone that stimulates cells to divide. EGF stimulates the growth of various epidermal and epithelial tissues in vivo and in vitro and of some fibroblasts in cell culture.1
EGF acts by binding with high affinity to epidermal growth factor receptor (EGFR) on the cell surface and stimulating the intrinsic protein-tyrosine kinase activity of the receptor. The tyrosine kinase activity, in turn, initiates a signal transduction cascade that results in a variety of biochemical changes within the cell, a rise in intracellular calcium levels, increased glycolysis and protein synthesis, and increases in the expression of certain genes including the gene for EGFR - that ultimately lead to DNA synthesis and cell proliferation.2
REFERENCES
1. Carpenter, G. & Cohen, S.:J. Biol. Chem. 265:7709-12, 1990
2. Eblin, K.: Toxicol. Sci.109:169-71, 2009
2. Eblin, K.: Toxicol. Sci.109:169-71, 2009
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10075 |
Antigen: | Purified recombinant human EGF expressed in HEK293 cells. |
Isotype: | Mouse IgG |
Species & predicted species cross- reactivity ( ): | Human, Mouse |
Applications & Suggested starting dilutions:* | WB 1:1000 IP 1:50 IHC 1:200 ICC 1:200 ELISA 1:10000 |
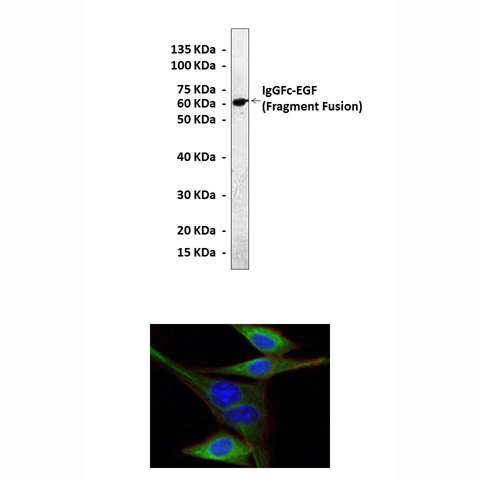
Predicted Molecular Weight of protein: | 6.2 kDa |
Specificity/Sensitivity: | Detects endogenous EGF proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой