Anti-FES: Mouse Feline Sarcoma Oncogene Antibody |
 |
BACKGROUND The human c-FES gene encodes a 93-kDa nonreceptor protein-tyrosine kinase (FES) that is predominantly expressed in myeloid hematopoietic cells, the vascular endothelium, and several types of neurons. Structurally, FES consists of a C-terminal kinase domain, a central SH2 domain, and a long N-terminal region. While the SH2 and kinase domains are closely related to those found in other cytoplasmic tyrosine kinases, such as c-Src and c-Abl, the N-terminal region is unique to FES and the closely related kinase Fer. It has been identified that at least two regions of coiled-coil motifs within the unique N-terminal contribute to the regulation of kinase activity in vivo. Unlike its transforming viral counterparts, FES tyrosine kinase activity is strictly regulated in mammalian cells. However, mutation or deletion of the more N-terminal coiled-coil motif is sufficient to release c-FES tyrosine kinase and transforming activities in rodent fibroblasts and to induce morphological differentiation of hematopoietic and neuronal cell lines.1 The N-terminal region also includes a FES/CIP4 homology (FCH) domain, which was first described as a region of homology between FES/Fer and the Cdc42-interacting protein, CIP4. The FCH domains of CIP4 and FES have been reported to bind to microtubules (MTs). Growing evidence suggests that MT stability may also be regulated by tyrosine phosphorylation.2 For example, tyrosine phosphorylation of tubulin by the Src family kinases Lyn and Fyn alters tubulin assembly and monocyte differentiation in the HL-60 myeloid leukemia cell line. It was shown that human FES tyrosine kinase binds and phosphorylates tubulin and microtubules through separate domains and promotes microtubule assembly. FES is a regulator of the tubulin cytoskeleton that may contribute to FES-induced morphological changes in myeloid hematopoietic and neuronal cells. In addition, many other FES-mediated signaling pathways were shown to be involved in hematopoitic cell differentiation. FES regulates the survival and differentiation of erythroid cells through modulation of Stat5A/B and Erk kinase pathways induced by EPO and SCF.3
REFERENCES
1. Smithgall, T.E. et al: Crt. Rev. Oncol. 9:43-62, 1998
2. Takashima, Y. et al: Biochem. 42:3567-74, 2003
3. Sangrar, W. et al: Exp. Hematol. 32:935-45, 2004
2. Takashima, Y. et al: Biochem. 42:3567-74, 2003
3. Sangrar, W. et al: Exp. Hematol. 32:935-45, 2004
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10100 |
Antigen: | Purified recombinant human FES fragments expressed in E. coli. |
Isotype: | Mouse IgG |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
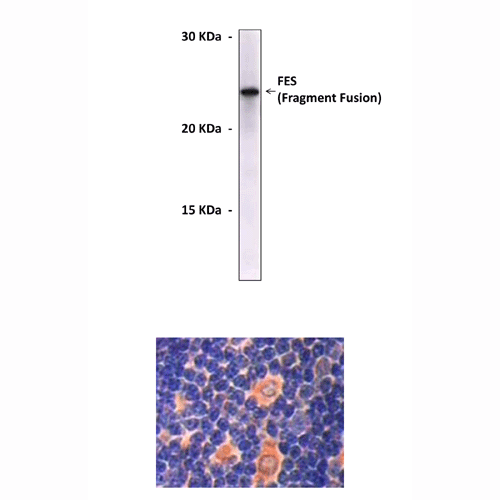
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:200 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 93 kDa |
Specificity/Sensitivity: | Detects FES proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой