Anti-Lamin-B: Rabbit Lamin-B Antibody |
 |
BACKGROUND The nuclear lamina, a protein meshwork that lines the inner nuclear membrane, is critical in fundamental cellular processes, including nuclear organization, chromatin segregation, DNA replication, and gene transcription. The principal protein components of the lamina are lamins, which are members of the intermediate filament protein family. Like other intermediate filament proteins, lamins possess an amino-terminal head domain and a highly conserved central α-rod domain for polymerization and oligomerization. Lamins are, however, distinguished from other intermediate filament proteins by a nuclear localization motif. In addition, prelamin A and lamins B1 and B2 contain a carboxyl-terminal CAAX motif that triggers a series of posttranslational modifications (farnesylation, endoproteolytic trimming of the last three amino acid residues, and methylation of the newly exposed farnesylcysteine). Aside from their structural role in the formation of the nuclear lamina, lamins A and C are found in the nucleoplasm adjacent to sites of DNA synthesis and RNA processing, suggesting that these proteins could influence both DNA replication and gene expression.1
In vertebrates, lamins are classified as A or B type, based on sequence homology, expression pattern, biochemical properties, and localization during mitosis. The A-type lamins, lamins A and C, are synthesized from alternatively spliced transcripts of LMNA and are expressed in most differentiated cells. Somatic cells also express two B-type lamins, lamin B1 and lamin B2, which are encoded by LMNB1 and LMNB2, respectively.2 Although the A- and B-type lamins interact with an overlapping set of other nuclear envelope proteins, they exhibit distinct expression patterns and different assembly properties, suggesting independent functions. For example, B-type lamins are expressed throughout development, whereas A-type lamins are expressed only after commitment of cells to a particular differentiation pathway. Also, A- and B-type lamins exhibit distinct solubility properties during mitosis because of temporal differences in the association with chromatin, with the association of B-type lamins preceding that of the A-type lamins. Mitotic spindle morphogenesis is a series of highly coordinated movements that lead to chromosome segregation and cytokinesis. It was demonstrated that lamin B assembled into a matrix-like network in mitosis through a process that depended on the presence of the guanosine triphosphate (GTP)-bound form of the small GTPase Ran. Depletion of lamin B resulted in defects in spindle assembly. Furthermore, lamin B was essential for the formation of the mitotic matrix that tethers a number of spindle assembly factors. Thus, it is suggested that lamin B is a structural component of the long-sought-after spindle matrix that promotes microtubule assembly and organization in mitosis.3 The lamin B receptor (LBR) is an integral membrane protein of the inner nuclear membrane that has so far been characterized only in vertebrates. The nucleoplasmic domain of LBR binds to B-type lamins, DNA, chromosomes and chromatin, and interacts with human chromodomain protein HP1 and nuclear membrane.4
In vertebrates, lamins are classified as A or B type, based on sequence homology, expression pattern, biochemical properties, and localization during mitosis. The A-type lamins, lamins A and C, are synthesized from alternatively spliced transcripts of LMNA and are expressed in most differentiated cells. Somatic cells also express two B-type lamins, lamin B1 and lamin B2, which are encoded by LMNB1 and LMNB2, respectively.2 Although the A- and B-type lamins interact with an overlapping set of other nuclear envelope proteins, they exhibit distinct expression patterns and different assembly properties, suggesting independent functions. For example, B-type lamins are expressed throughout development, whereas A-type lamins are expressed only after commitment of cells to a particular differentiation pathway. Also, A- and B-type lamins exhibit distinct solubility properties during mitosis because of temporal differences in the association with chromatin, with the association of B-type lamins preceding that of the A-type lamins. Mitotic spindle morphogenesis is a series of highly coordinated movements that lead to chromosome segregation and cytokinesis. It was demonstrated that lamin B assembled into a matrix-like network in mitosis through a process that depended on the presence of the guanosine triphosphate (GTP)-bound form of the small GTPase Ran. Depletion of lamin B resulted in defects in spindle assembly. Furthermore, lamin B was essential for the formation of the mitotic matrix that tethers a number of spindle assembly factors. Thus, it is suggested that lamin B is a structural component of the long-sought-after spindle matrix that promotes microtubule assembly and organization in mitosis.3 The lamin B receptor (LBR) is an integral membrane protein of the inner nuclear membrane that has so far been characterized only in vertebrates. The nucleoplasmic domain of LBR binds to B-type lamins, DNA, chromosomes and chromatin, and interacts with human chromodomain protein HP1 and nuclear membrane.4
REFERENCES
1. Gruenbaum, Y. et al: J. Struct. Biol. 129:313-23, 2000
2. Goldberg, M. et al: Crit. Rev. Eukaryot. Gene Expr. 9:285-93, 1999
3. Tsai, M.Y. et al: Science 311:1887-93,2006
4. Wagner, N. et al: J. Cell Sci. 117:2015-28, 2004
2. Goldberg, M. et al: Crit. Rev. Eukaryot. Gene Expr. 9:285-93, 1999
3. Tsai, M.Y. et al: Science 311:1887-93,2006
4. Wagner, N. et al: J. Cell Sci. 117:2015-28, 2004
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CA1048 |
Antigen: | Short peptide from human Lamin-B sequence. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Rat |
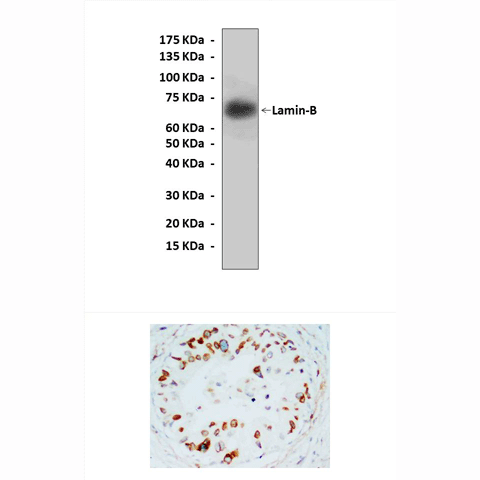
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:50 - 1:200 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 70 kDa |
Specificity/Sensitivity: | Detects endogenous levels of lamin-B proteins without cross-reactivity with other related proteins. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой