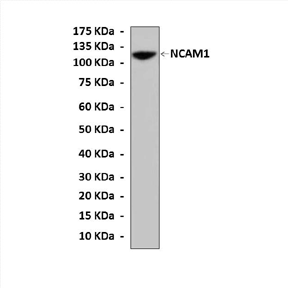
Anti-NCAM1: Rabbit Neural Cell Adhesion Molecule Antibody |
 |
BACKGROUND The neural cell adhesion molecule (NCAM1, aliases NCAM and CD56) is a multifunction transmembrane protein involved in synaptic plasticity, neurodevelopment, and neurogenesis. NCAM1 is found on chromosome 11q23.1 and is part of the immunoglobin superfamily. The extracellular domain of NCAM1 consists of five Ig modules (IgI–V) and two fibronectin type III homology modules (F3, I and II). Importantly, NCAM1 promotes neurite outgrowth via homophilic (NCAM1–NCAM1) as well as heterophilic (NCAM1–FGFR, other CAMs and ECM proteins) interactions, which activate a number of intracellular signaling cascades. Recently, a novel mechanism of homophilic NCAM1 binding has been proposed, in which the IgI and IgII modules mediate dimerization of NCAM molecules situated on the same cell surface (cis interactions), whereas trans interactions between the IgI and IgIII, IgII and IgII, and the IgII and IgIII modules of NCAM1 determine cell–cell adhesion.1
NCAM1 is expressed in both neurons and glial cells with cell recognition properties involved in cellular migration, synaptic plasticity and central nervous system development. Three major isoforms of NCAM1 (180, 140, 120) arise from a single copy gene via RNA processing, specifically alternative splicing and polyadenylation, whereas NCAM1, 105–115 kDa, appears to result from posttranslational modifications. NCAM1 involves several alternatively spliced exons such as the variable alternative spliced exon (VASE) and the secreted exon (SEC), which are expressed in brain and appear to be developmentally regulated. Many other splice patterns have been identified in NCAM1 and if they were to be translated, it is predicted that they could result in up to 192 different NCAM1 proteins. In particular, the secreted isoform of NCAM1 (SECNCAM1) is increased in the hippocampus of BPD patients whereas a proteolytic cleavage isoform of NCAM (cNCAM) was not altered in the brain of patients with BPD but increased in SZ. Transgenic NCAM1 mice show some suggestive parallels to SZ such as alterations in brain ventricular ratio and alterations in prepulse inhibition. Additionally, a recent mouse model that overexpresses a truncated form of NCAM1 similar to the cleaved form of NCAM1 in human neuropsychiatric disorders also exhibits alterations in synaptophysin, GABAergic neurons, and behavior.2
In addition to its role in cell adhesion, NCAMs act as a signal receptor molecule. It was shown that NCAM1 also induces activation of a complex network of intracellular signaling cascades on homophilic or heterophilic binding. Stimulation of NCAM1 by homophilic interactions induces neuronal differentiation through activation of a number of signaling molecules, including the fibroblast growth factor receptor, non–receptor kinases Fyn and focal adhesion kinase, growth-associated protein-43, the mitogen-activated protein kinase pathway, intracellular Ca2+, and protein kinases A, C, and G.3
NCAM1 is expressed in both neurons and glial cells with cell recognition properties involved in cellular migration, synaptic plasticity and central nervous system development. Three major isoforms of NCAM1 (180, 140, 120) arise from a single copy gene via RNA processing, specifically alternative splicing and polyadenylation, whereas NCAM1, 105–115 kDa, appears to result from posttranslational modifications. NCAM1 involves several alternatively spliced exons such as the variable alternative spliced exon (VASE) and the secreted exon (SEC), which are expressed in brain and appear to be developmentally regulated. Many other splice patterns have been identified in NCAM1 and if they were to be translated, it is predicted that they could result in up to 192 different NCAM1 proteins. In particular, the secreted isoform of NCAM1 (SECNCAM1) is increased in the hippocampus of BPD patients whereas a proteolytic cleavage isoform of NCAM (cNCAM) was not altered in the brain of patients with BPD but increased in SZ. Transgenic NCAM1 mice show some suggestive parallels to SZ such as alterations in brain ventricular ratio and alterations in prepulse inhibition. Additionally, a recent mouse model that overexpresses a truncated form of NCAM1 similar to the cleaved form of NCAM1 in human neuropsychiatric disorders also exhibits alterations in synaptophysin, GABAergic neurons, and behavior.2
In addition to its role in cell adhesion, NCAMs act as a signal receptor molecule. It was shown that NCAM1 also induces activation of a complex network of intracellular signaling cascades on homophilic or heterophilic binding. Stimulation of NCAM1 by homophilic interactions induces neuronal differentiation through activation of a number of signaling molecules, including the fibroblast growth factor receptor, non–receptor kinases Fyn and focal adhesion kinase, growth-associated protein-43, the mitogen-activated protein kinase pathway, intracellular Ca2+, and protein kinases A, C, and G.3
REFERENCES
1. Kiselyov, V. et al: Structure 11:691-701, 2003
2. Sytnyk, V. et al: J. Cell Biol. 174:1071-85, 2006
3. Chekhonin,V.P. et al: Neurochem. J.1:113-26, 2007
2. Sytnyk, V. et al: J. Cell Biol. 174:1071-85, 2006
3. Chekhonin,V.P. et al: Neurochem. J.1:113-26, 2007
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CA1055 |
Antigen: | Short peptide from human NCAM1 sequence. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:50 - 1:200 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 105-180 kDa |
Specificity/Sensitivity: | Detects endogenous levels of NCAM1 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой