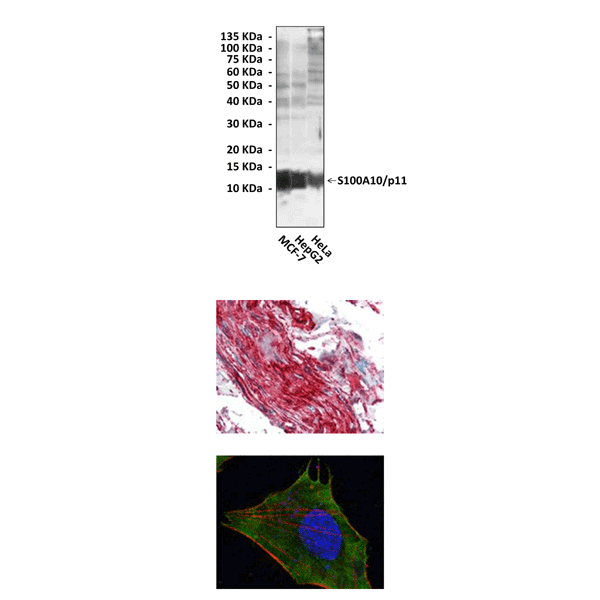
Anti-S100A10/p11: Mouse S100A10/p11 Antibody |
 |
BACKGROUND S100A10, also known as p11 or annexin 2 light chain, is a member of the S100 family of small, dimeric EF hand-type Ca2+-binding proteins that generally modulate cellular target proteins in response to intracellular Ca2+ signals. In contrast to all other S100 proteins, S100A10 is Ca2+ insensitive because of amino acid replacements in its Ca2+-binding loops that lock the protein in a permanently active state. Within cells, the majority of S100A10 resides in a tight heterotetrameric complex with the peripheral membrane-binding protein annexin A2 (AnxA2) that directs the complex to specific target membranes, in particular the plasma membrane and the membrane of early endosomes.1 AnxA2 is composed of a conserved C-terminus core domain folded via annexin repeats and a hypervariable non-folded N-terminus. AnxA2 N-terminal domain is small (24 amino acids) and bears two putative phosphorylation sites Tyr23 and Ser25, which are presumably targets of Src kinase and protein kinase C, respectively, as well as the binding site for its natural ligand p11/S100A10. Interactions of two molecules of AnxA2 with two molecules of p11 lead to the formation of the (AnxA2)2-(p11)2 heterotetramer. It has been reported that the p11 light chain is required for AnxA2 binding to the plasma membrane and to the cortical actin network, both mechanisms being also regulated by the presence of Ca2+. Evidence also suggests that the (AnxA2)2-(p11)2 heterotetramer plays a role in the subcellular distribution of early and recycling endosomes and in the channel functions of cystic fibrosis conductance regulator protein.2 Several other Ca2+-independent interaction partners of S100A10 have been described in the recent past. Many of these interactions, which have been shown to be of functional significance for the respective partner, involve plasma membrane-resident proteins. In most of these cases, S100A10, probably residing in a complex with annexin A2, appears to regulate the intracellular trafficking of the respective target protein and thus its functional expression at the cell surface. It is suggested that S100A10 functions as a linker tethering certain transmembrane proteins to annexin A2 thereby assisting their traffic to the plasma membrane and/or their firm anchorage at certain membrane sites.3
REFERENCES
1. Svenningsson, P. & Greengard, P:Curr. Opin. Pharmcol. 7:27-32, 2007
2. Morel, E. & Gruenberg, J.:PLOS ONE 2:e1118, 2007
3. Rescher, U. & Volker, Gerke. V.: Pflügers Archiv Eur. J. Physiol. 455:575-82, 2007
2. Morel, E. & Gruenberg, J.:PLOS ONE 2:e1118, 2007
3. Rescher, U. & Volker, Gerke. V.: Pflügers Archiv Eur. J. Physiol. 455:575-82, 2007
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10222 |
Antigen: | Purified recombinant human S100A10/p11 protein expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:200 ICC 1:200 FACS n/d |
Predicted Molecular Weight of protein: | 11 kDa |
Specificity/Sensitivity: | Detects endogenous S100A10/p11 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой