RANK/TNFRSF11A ELISA Kit, Human |
 |
BACKGROUND RANK (receptor activator of nuclear factor‐κB also known as TRANCE-R, ODAR, or TNFRSF11A) is a member of TNF-R superfamily. It is the receptor for RANKL. The tumor necrosis factor family molecule RANKL (RANKL, TRANCE, ODF) and its receptor RANK are key regulators of bone remodeling and regulate T cell/ dendritic cell communications, and lymph node formation.1 Moreover, RANKL and RANK are expressed in mammary gland epithelial cells and control the development of a lactating mammary gland during pregnancy and the propagation of mammalian species. Importantly, RANKL and RANK are essential for the development and activation of osteoclasts and bone loss in response to virtually all triggers tested. RANK is expressed as a transmembrane heterotrimer on the surface of haematopoietic osteoclasts progenitors, mature osteoclasts, chondrocytes, mammary gland epithelial cells, trophoblast cells, and multiple epithelial tumor cell lines. In vitro ligation of RANK with RANKL results in osteoclastogenesis from progenitor cells and the activation of mature osteoclasts. The interaction between RANKL expressed by stromal cells/osteoblasts and its receptor RANK expressed on osteoclast precursors are essential for osteoclastogenesis. Mice with a genetic mutation of RANK are phenocopies of rankl-/- mice and have a complete block in osteoclast development that can be restored by reintroduction of RANK into bone marrow progenitor cells. In human, RANK mutations cause Paget's disease of bone (PDB)–like diseases (ie, familial expansile osteolysis, expansile skeletal hyperphosphatasia, and early‐onset PDB) and an osteoclast‐poor form of osteopetrosis. All biochemical, functional, and genetic results in humans and mutant mice established the absolute dependency of osteoclast differentiation and activation of mature osteoclasts on the expression of RANKL and RANK.2
When RANK on osteoclasts is activated it sends signals into the cells through adapter proteins. RANK contains 383 amino acids in its intracellular domain (residues 234–616), which contain three putative binding domains (termed I, II, and III) for tumor necrosis factor receptor associated factors (TRAFs). These TRAF binding domains were shown to be functionally important for the RANK dependent induction of NF- κB and JNK activities. However, interaction with TRAFs is necessary for NFκB activation but not essential for activation of the JNK pathway. RANKL also activates the anti-apoptotic Akt through a signaling complex involving c-Src and TRAF6. c-Src and TRAF6 interact with each other and with RANK after receptor engagement and a deficiency in c-Src or addition of Src family kinase inhibitors blocks TRANCE mediated Akt activation in osteoclasts. TRAF6, in turn, increases the kinase activity of c-Src leading to tyrosine phosphorylation of downstream signaling molecules such as c-Cbl. Moreover, RANK can recruit TRAF6, Cbl family scaffolding proteins, and the phospholipid kinase PI3-K in a ligand and Src dependent manner. RANKL mediated Akt activation is defective in cbl-b-/- dendritic cells. Thus, Cbl family proteins are not only negative regulators of signaling, but also the positive modulators of TNFR superfamily signalling.3
When RANK on osteoclasts is activated it sends signals into the cells through adapter proteins. RANK contains 383 amino acids in its intracellular domain (residues 234–616), which contain three putative binding domains (termed I, II, and III) for tumor necrosis factor receptor associated factors (TRAFs). These TRAF binding domains were shown to be functionally important for the RANK dependent induction of NF- κB and JNK activities. However, interaction with TRAFs is necessary for NFκB activation but not essential for activation of the JNK pathway. RANKL also activates the anti-apoptotic Akt through a signaling complex involving c-Src and TRAF6. c-Src and TRAF6 interact with each other and with RANK after receptor engagement and a deficiency in c-Src or addition of Src family kinase inhibitors blocks TRANCE mediated Akt activation in osteoclasts. TRAF6, in turn, increases the kinase activity of c-Src leading to tyrosine phosphorylation of downstream signaling molecules such as c-Cbl. Moreover, RANK can recruit TRAF6, Cbl family scaffolding proteins, and the phospholipid kinase PI3-K in a ligand and Src dependent manner. RANKL mediated Akt activation is defective in cbl-b-/- dendritic cells. Thus, Cbl family proteins are not only negative regulators of signaling, but also the positive modulators of TNFR superfamily signalling.3
REFERENCES
1. Baud'huin, M. et al: Expert Rev. Anticancer Ther. 7:221-32, 2007
2. Boyce, B.F. & Xing, L.: Curr. Osteoporos. Rep. 5:98-104, 2007
3. Boyce, B.F. & Xing, L.: Arch. Biochem. Biophys. 473:139-46, 2008
2. Boyce, B.F. & Xing, L.: Curr. Osteoporos. Rep. 5:98-104, 2007
3. Boyce, B.F. & Xing, L.: Arch. Biochem. Biophys. 473:139-46, 2008
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CL0829 |
Target Protein Species: | Human |
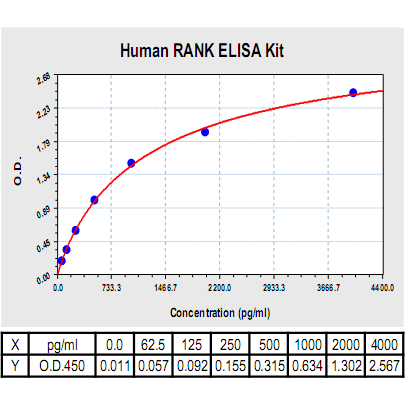
Range: | 62.5pg/ml-4000pg/ml |
Specificity: | No detectable cross-reactivity with any other cytokine. |
Storage: | Store at 4°C. Use within 6 months. |
ELISA Kits are based on standard sandwich enzyme-linked immunosorbent assay technology. Freshly prepared standards, samples, and solutions are recommended for best results.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой