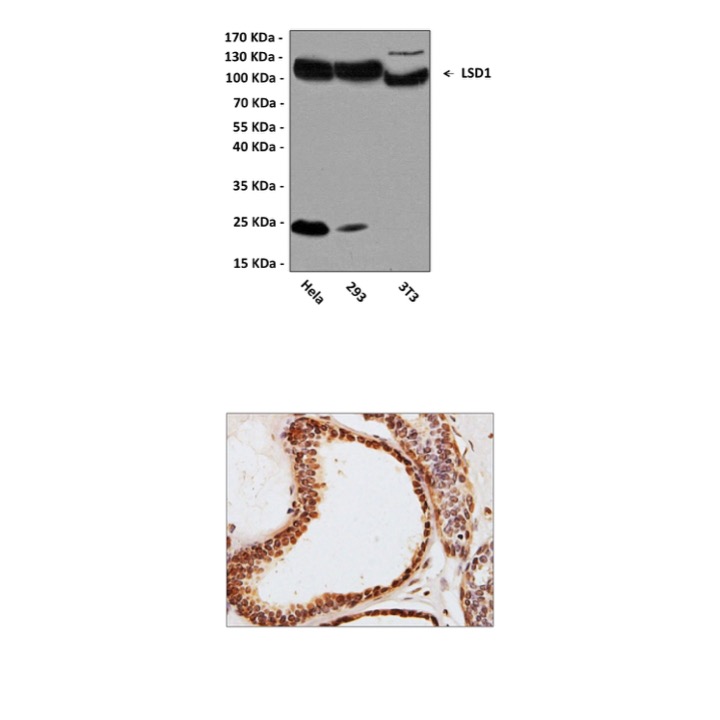
Anti-LSD1: Mouse LSD1 Antibody |
 |
BACKGROUND Lysine-specific demethylase 1 (LSD1; also known as AOF2 and BHC110) is a nuclear homolog of amine oxidases and functions as a histone demethylase and transcription cofactor (1). Gene activation and repression is specifically regulated by the methylation state of distinct lysine residues in histone proteins. For example, methylation of histone H3 Lys4 facilitates transcriptional activation by coordinating the recruitment of BPTF, a component of the NURF chromatin remodeling complex, and WDR5, a component of multiple histone methyltransferase complexes (2, 3). In contrast, methylation of histone H3 (Lys9) facilitates transcriptional repression by recruiting HP1 (4, 5). LSD1 is a component of the CoREST transcriptional co-repressor complex, which contains CoREST, CtBP, HDAC1 and HDAC2. As part of this complex, LSD1 demethylates mono-methyl and di-methyl histone H3 Lys4 via an FADdependent oxidation reaction, and facilitates the repression of neuronal-specific genes in non-neuronal cells (1, 6, 7). In contrast, when LSD1 is associated with androgen-receptor in human prostate cells, it demethylates mono-methyl and di-methyl histone H3 Lys9 and facilitates androgen-receptor dependent transcriptional activation (8). Therefore, LSD1 can function as a co-repressor or co-activator, depending on gene context. LSD1 activity is inhibited by the amine oxidase inhibitors pargyline, deprenyl, clorgyline and tranylcypromine (8).
REFERENCES
1. Shi, Y. et al. (2004) Cell 119, 941–953.
2. Wysocka, J. et al. (2006) Nature 442, 86–90.
3. Wysocka, J. et al. (2005) Cell 121, 859–872.
4. Jacobs, S.A. and Khorasanizadeh, S. (2002) Science295, 2080–2083.
5. Nielsen, P.R. et al. (2002) Nature 416, 103–107.
6. Shi, Y.J. et al. (2005) Mol. Cell 19, 857–864.
7. Lee, M.G. et al. (2005) Nature 437, 432–435.
8. Metzger, E. et al. (2005) Nature 437, 436–439.
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
| Cat.No.: | CC10052 |
| Antigen: | Recombinant human LSD1 fragment expressed in E. Coli |
| Isotype: | Mouse IgG |
Species & predicted species cross- reactivity ( ): | Human, Mouse, |
Applications & Suggested starting dilutions:* | WB 1:10000 - 1:50000 IP n/d IHC 1:100 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 110 kDa |
| Specificity/Sensitivity: | Detects endogenous LSD1 proteins without cross-reactivity with other family members. |
| Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой