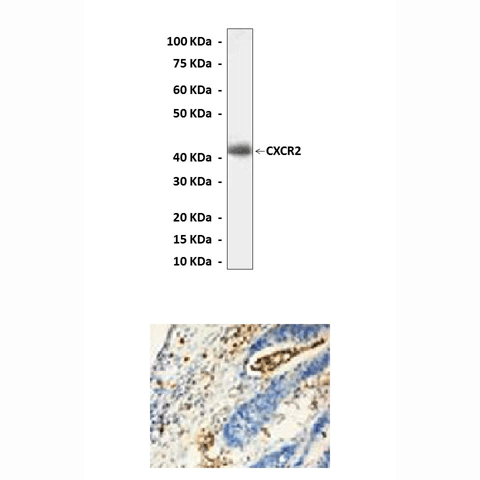
Anti-CXCR2: Polyclonal CXCR2 Antibody |
 |
BACKGROUND Chemokines induce cell migration and activation by binding to specific seven transmembrane (7TM) G protein-coupled cell surface receptors (GPCRs) on target cells. Chemokine receptors, like all members of the GPCR superfamily, mediate signal transduction through G proteins. Two receptors for chemokine CXCL8 (IL-8), the type A CXCL8 receptor (CXCL8RI or CXCR1) and type B CXCL8 receptor (CXCL8RII or CXCR2) have been shown to bind CXCL8 with high affinity. CXCR1 is selective for CXCL8, whereas CXCR2 also interacts with other chemokines. They are widely co-expressed on immune cells, including neutrophils, CD8(+) T cells, and mast cells; as well as on keratinocytes, fibroblasts, and endothelial and melanoma cells. Moreover, it was shown that both CXCR1 and CXCR2 form homodimer/oligomers at an early stage in synthesis and maturation and that when co-expressed CXCR1 and CXCR2 form heterodimers as effectively as homodimers. The extent of neither the CXCR1-CXCR2 heterodimer nor the corresponding homodimers is affected by the presence of IL8.1 However, It was also found that low expression of CXCR2 on normal human melanocytes, which was up-regulated after treatment with tumor necrosis factor alpha, with subsequent enhancement of proliferation in response to CXCL8, whereas CXCR1 expression was not detectable.2
CXCL8, in addition to other cytokines such has IL-6, has been implicated in tissue inflammation and repair as well as carcinogenesis. It has been shown to be an important angiogenic factor. Both CXCR1 and CXCR2 also have been implicated in the angiogenic response and in the migration of neutrophils and lymphocytes. It has been demonstrated that Phosphatidylinositol-3 kinase (PI3K), the GTP-binding proteins Rac, Rho and cdc42 as well as phospholipase c participate in regulating CXCR1-mediated chemotaxis, and Cbl and Akt are also key components of this pathway.3 Moreover, CXCL8/CXCR1/2 may be involved in regulation of self-renewal or survival pathways in cancer stem cell (CSC). CXCR1 activation upon IL-8 binding induces FAK phosphorylation. Active FAK phosphorylates AKT and activates the WNT pathway, which regulates stem cell self-renewal and FOXO3A that regulates cell survival. Activation of FAK protects CSCs from a FASL/FAS-mediated bystander effect by inhibiting FADD, a downstream effector of FAS signaling. In the presence of chemotherapy, only the bulk tumor cells are sensitive to the treatment and release a high level of IL-8 and FASL proteins during the apoptotic process. Breast CSCs are stimulated via an IL-8–mediated bystander effect and are resistant to the bystander killing effect mediated by FASL. CXCR1 blockade may provide a novel means of targeting and eliminating breast CSCs.4
CXCL8, in addition to other cytokines such has IL-6, has been implicated in tissue inflammation and repair as well as carcinogenesis. It has been shown to be an important angiogenic factor. Both CXCR1 and CXCR2 also have been implicated in the angiogenic response and in the migration of neutrophils and lymphocytes. It has been demonstrated that Phosphatidylinositol-3 kinase (PI3K), the GTP-binding proteins Rac, Rho and cdc42 as well as phospholipase c participate in regulating CXCR1-mediated chemotaxis, and Cbl and Akt are also key components of this pathway.3 Moreover, CXCL8/CXCR1/2 may be involved in regulation of self-renewal or survival pathways in cancer stem cell (CSC). CXCR1 activation upon IL-8 binding induces FAK phosphorylation. Active FAK phosphorylates AKT and activates the WNT pathway, which regulates stem cell self-renewal and FOXO3A that regulates cell survival. Activation of FAK protects CSCs from a FASL/FAS-mediated bystander effect by inhibiting FADD, a downstream effector of FAS signaling. In the presence of chemotherapy, only the bulk tumor cells are sensitive to the treatment and release a high level of IL-8 and FASL proteins during the apoptotic process. Breast CSCs are stimulated via an IL-8–mediated bystander effect and are resistant to the bystander killing effect mediated by FASL. CXCR1 blockade may provide a novel means of targeting and eliminating breast CSCs.4
REFERENCES
1. Wilson, S. Et al: J. Biol. Chem. 280:28663-74, 2005
2. Varney, M.L. et al: Am. J. Clin. Pathol. 125:209-16, 2006
3. Lane, H.C. et al: Int. Immunol. 18:1315-25, 2006
4. Ginestier, C. et al: J. Clin. Invest. 120:485–97, 2010
2. Varney, M.L. et al: Am. J. Clin. Pathol. 125:209-16, 2006
3. Lane, H.C. et al: Int. Immunol. 18:1315-25, 2006
4. Ginestier, C. et al: J. Clin. Invest. 120:485–97, 2010
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CA0757 |
Antigen: | Central part sequence of human CXCR2 |
Isotype: | Affinity-purified rabbit polyclonal IgG |
Species & predicted species cross- reactivity ( ): | Human, Rabbit, Rat, Mouse |
Applications & Suggested starting dilutions: | WB 1:500 to 1:1000 IP n/d IHC (Paraffin) 1:50 to 1:200 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 42 kDa |
Specificity/Sensitivity: | No cross-reactivity with other members of the family. |
Storage: | Store at 4° C for frequent use; at -20° C for at least one year. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой