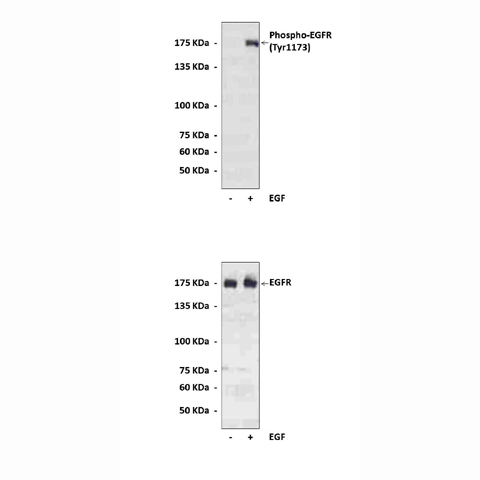
Anti-Phospho-EGFR: Polyclonal Epidermal Growth Factor Receptor, Phospho-Tyr1173 Antibody |
 |
BACKGROUND The epidermal growth factor receptor (EGFR) is a transmembrane glycoprotein that constitutes one of four members of the erbB family of tyrosine kinase receptors.1 Upon activation by at least five genetically distinct ligands (including EGF, transforming growth factor-α (TGFα) and heparin-binding EGF (HB-EGF), the intrinsic kinase is activated and EGFR tyrosyl-phosphorylates itself and numerous intermediary effector molecules, including closely-related c-erbB receptor family members. This initiates myriad signaling pathways. The integrated biological responses to EGFR signaling are pleiotropic including mitogenesis or apoptosis, enhanced cell motility, protein secretion, and differentiation or dedifferentiation.2 Although present in normal cells, EGFR is overexpressed in a variety of tumor cell lines and has been associated with poor prognosis and decreased survival. EGFR activation also plays a role in resistance to chemotherapy and radiation treatment in tumor cells.3 Thus, EGFR and its downstream signaling molecules are targets for therapeutic interventions in cancer and many other clinical pathogenesis.4
REFERENCES
1. Burgess AW: Growth Factors, 26:263-274, 2008.
2. Normanno N et al.: Gene 366:2-16, 2006.
3. Hopper-Borge EA et al.: Expert Opin Ther Targets. 13:339-362, 2009.
4. Sharma PS et al.: Curr Pharm Des. 15:758-776, 2009.
2. Normanno N et al.: Gene 366:2-16, 2006.
3. Hopper-Borge EA et al.: Expert Opin Ther Targets. 13:339-362, 2009.
4. Sharma PS et al.: Curr Pharm Des. 15:758-776, 2009.
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CC1049 |
Antigen: | Sequence surrounding and including Tyr1173 of human EGFR . |
Isotype: | Affinity purified rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions: | WB 1:1000 IP n/d IHC (Paraffin) n/d ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 175 kDa |
Specificity/Sensitivity: | Detects endogenous phospho-human, mouse and rat EGFR proteins. |
Storage: | Store at 4° C for frequent use; at -20° C for at least one year. |
*Optimal working dilutions must be determined by end user.
Документы
|
| ||||||||||
| ||||||||||
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой