Anti-GSK-3α: Mouse Glycogen Synthase Kinase 3 alpha Antibody |
 |
BACKGROUND Glycogen Synthase Kinase 3 (GSK-3), a ubiquitously expressed and evolutionarily conserved protein serine/threonine kinase, was originally identified as an enzyme that regulates glycogen synthesis in response to insulin. GSK-3 was subsequently shown to function in cell division, proliferation, motility and survival. GSK-3 plays a role in a number of pathological conditions including cancer and diabetes and is increasingly seen as an important component of neurological diseases. GSK-3 phosphorylates tau and presenilin-1, which are involved in the development of Alzheimer's disease. Both isoforms of GSK-3 are ubiquitously expressed, although particularly high levels of GSK-3beta are found in the brain where it is involved in synaptic plasticity, possibly via regulation of NMDA receptor trafficking.1
There are two mammalian GSK-3 isoforms encoded by distinct genes: GSK-3alpha and GSK-3beta. They are closely related in function. GSK-3alpha has a mass of 51 kDa, whereas GSK-3beta is a protein of 47 kDa. The difference in size is due to a glycine-rich extension at the N-terminus of GSK-3alpha. Although highly homologous within their kinase domains (98% identity), the two gene products share only 36% identity in the last 76 C-terminal residues. Moreover, they are not functionally identical. GSK-3 phosphorylates over 40 different substrates including signaling proteins, transcription factors and structural proteins, and is part of the signal transduction cascade of a large number of growth factors and cytokines. GSK-3 has also been implicated in the regulation of cell fate in Dictyostelium and is a component of the Wnt signaling pathway required for Drosophila and Xenopus development.2
The activity of GSK is regulated by phosphorylation (Akt, S6K, RSK, PKA and PKC), dephosphorylation (PP1 and PP2A), and by binding to protein complexes (with beta-catenin, axin, CK1 and the APC complex). In mammalian cells, on stimulation with insulin or other growth factors, GSK-3 is rapidly phosphorylated at serine 21 in GSK-3alpha or serine 9 in GSK-3beta, resulting in inhibition of GSK-3 kinase activity. Protein kinase B (PKB/Akt), a serine/threonine kinase located downstream of Phosphatidylinositol 3-kinase (PI3K), has been demonstrated to phosphorylate both of these sites in vitro and in vivo, suggesting that growth factors down-regulate GSK-3 activity through the PI3K–PKB signaling cascade. Consistent with its position downstream of the PI3K–PKB pathway, GSK-3 activity suppresses cell proliferation and survival.3 It was also demonstrated that serine 21 in GSK-3alpha and Serine 9 in GSK-3beta are also physiological substrates of cAMP-dependent Protein Kinase A (PKA). PKA physically associates with, phosphorylates, and inactivates both isoforms of GSK-3. Thus PKA functions as a GSK-3 kinase that, in parallel with PKB, controls the activity of the multifunctional enzyme GSK-3.4
REFERENCES
1. Ali, A. et al: Chem. Rev. 101:2527–40, 2001
2. Doble, B.W. & Woodgett, J.R.: J. Cell Sci. 116:1175-86, 2003
3. Elder-Finkelman, H. et al: Trends Mol. Med. 8:126-32, 2002
4. Fang, X. et al: Proc. Natl. Acad. Sci. USA 97:11960-5, 2000
2. Doble, B.W. & Woodgett, J.R.: J. Cell Sci. 116:1175-86, 2003
3. Elder-Finkelman, H. et al: Trends Mol. Med. 8:126-32, 2002
4. Fang, X. et al: Proc. Natl. Acad. Sci. USA 97:11960-5, 2000
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10118 |
Antigen: | Purified recombinant human GSK-3 alpha fragments expressed in E. coli. |
Isotype: | Mouse IgG |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC n/d FACS n/d |
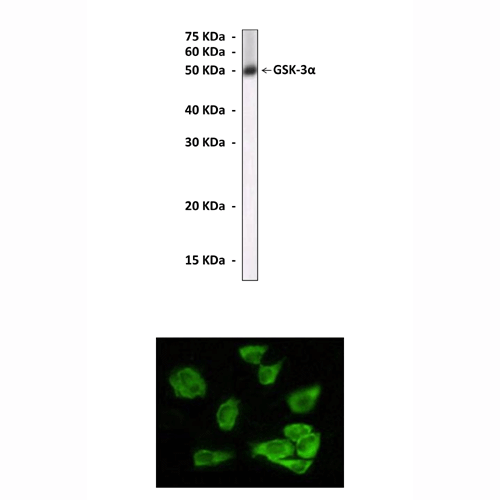
Predicted Molecular Weight of protein: | 51 kDa |
Specificity/Sensitivity: | Detects endogenous GSK-3 alpha proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой