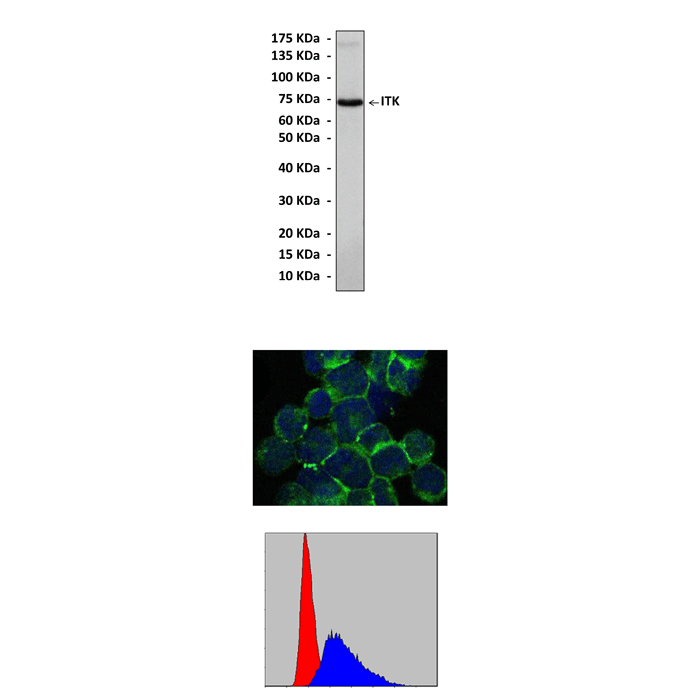
Anti-ITK: Mouse ITK Antibody |
 |
BACKGROUND The Tec family nonreceptor tyrosine kinases, Tec, Btk, Itk/Emt/Tsk, Rlk/Txk, and Bmx/Etk, are expressed primarily in hematopoietic cells and serve as important mediators of antigen receptor signaling in lymphocytes. Itk (inducible T-cell kinase; also known as Emt, expressed in mast cells and T lymphocytes, and Tsk, T-cell-specific kinase) was cloned in the early 1990s. The domain organization of Itk and related Tec kinase family members shares similarities with other tyrosine kinase families but also reveals unique features. Shared features include the common SH3-SH2-kinase cassette also found in the Src, Csk, and Abl kinases. Four of the five Tec tyrosine kinases (Itk, Btk, Tec, and Bmx) contain an amino-terminal region that includes a pleckstrin homology (PH) domain, followed by a Zn++ binding region termed the Btk homology (BH) motif, and (except for Bmx) a proline rich region (PRR) that conforms to the consensus sequence of an SH3 ligand.1
Although T lymphocytes express three Tec kinases: Itk, Rlk and Tec. To date, only Itk has been found to have a clearly defined function in T cells, leading to the conclusion that Itk is the predominant Tec kinase in T cells. Itk plays important role in activation via T-cell receptor (TCR) stimulation. Interaction of the TCR with peptide-MHC complexes on antigen presenting cells (APC) activates the Src kinase Lck, leading to phosphorylation of the CD3 immunoreceptor tyrosine-based activation motifs (ITAMs). Zap-70 then binds to the phosphorylated ITAMs and is phosphorylated by Lck, resulting in Zap-70 activation and subsequent phosphorylation of the adaptors LAT and SLP-76. Following activation of PI3K and accumulation of phosphatidylinositol (3,4,5) trisphosphate (PIP3) in the plasma membrane, Itk is recruited to the membrane via its PH domain. There, Itk interacts with the phosphorylated SLP-76/LAT adapter complex via Itk’s Src homology (SH3 and SH2) domains, permitting phosphorylation of Itk on its activation loop (Y511) by Lck. Once activated, Itk undergoes cis autophosphorylation on Y180 in its SH3 domain. Itk also interacts with and directly phosphorylates its downstream target, PLCγ1. Because autophosphorylation on Y180 does not affect Itk catalytic activity but instead modulates binding of the Itk SH3 domain to different targets, Itk autophosphorylation may serve as a trigger to alter protein interaction partners or localization either before or after PLCγ1 activation. PLCγ1, once activated, hydrolyzes PIP2 to produce the second messengers IP3 and DAG. Downstream consequences include Ca2+ flux, Erk activation, transcription, cytokine release, and actin reorganization, all of which are impaired in the absence of Itk. Importantly, these downstream consequences of TCR activation are not ablated by a deficiency in Itk, but instead, are substantially reduced, leading to altered development and differentiation of distinct T-cell lineages. In addition, it was demonstrated that there is a role for Itk in the development of invariant NKT αβ cells, as well as a smaller population NKT-like γδ T cells.2 Due to its critical role in acting as a modulator of T-cells, ITK inhibitors could provide a novel route to anti-inflammatory therapy.3
Although T lymphocytes express three Tec kinases: Itk, Rlk and Tec. To date, only Itk has been found to have a clearly defined function in T cells, leading to the conclusion that Itk is the predominant Tec kinase in T cells. Itk plays important role in activation via T-cell receptor (TCR) stimulation. Interaction of the TCR with peptide-MHC complexes on antigen presenting cells (APC) activates the Src kinase Lck, leading to phosphorylation of the CD3 immunoreceptor tyrosine-based activation motifs (ITAMs). Zap-70 then binds to the phosphorylated ITAMs and is phosphorylated by Lck, resulting in Zap-70 activation and subsequent phosphorylation of the adaptors LAT and SLP-76. Following activation of PI3K and accumulation of phosphatidylinositol (3,4,5) trisphosphate (PIP3) in the plasma membrane, Itk is recruited to the membrane via its PH domain. There, Itk interacts with the phosphorylated SLP-76/LAT adapter complex via Itk’s Src homology (SH3 and SH2) domains, permitting phosphorylation of Itk on its activation loop (Y511) by Lck. Once activated, Itk undergoes cis autophosphorylation on Y180 in its SH3 domain. Itk also interacts with and directly phosphorylates its downstream target, PLCγ1. Because autophosphorylation on Y180 does not affect Itk catalytic activity but instead modulates binding of the Itk SH3 domain to different targets, Itk autophosphorylation may serve as a trigger to alter protein interaction partners or localization either before or after PLCγ1 activation. PLCγ1, once activated, hydrolyzes PIP2 to produce the second messengers IP3 and DAG. Downstream consequences include Ca2+ flux, Erk activation, transcription, cytokine release, and actin reorganization, all of which are impaired in the absence of Itk. Importantly, these downstream consequences of TCR activation are not ablated by a deficiency in Itk, but instead, are substantially reduced, leading to altered development and differentiation of distinct T-cell lineages. In addition, it was demonstrated that there is a role for Itk in the development of invariant NKT αβ cells, as well as a smaller population NKT-like γδ T cells.2 Due to its critical role in acting as a modulator of T-cells, ITK inhibitors could provide a novel route to anti-inflammatory therapy.3
REFERENCES
1. Antherly, L.O. et al: Immunity 25:79-91, 2006
2. Qi, Q. et al: FEBS J. 278:1970-9, 2011
3. Cook, B.N. et al: Bioorganic Med. Chem. Lett. 19:773–7, 2009
2. Qi, Q. et al: FEBS J. 278:1970-9, 2011
3. Cook, B.N. et al: Bioorganic Med. Chem. Lett. 19:773–7, 2009
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10419 |
Antigen: | Raised against recombinant human ITK fragments expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC 1:50 - 1:200 FACS 1:50 - 1:200 |
Predicted Molecular Weight of protein: | 72 kDa |
Specificity/Sensitivity: | Detects ITK proteins in various cell lysate. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой