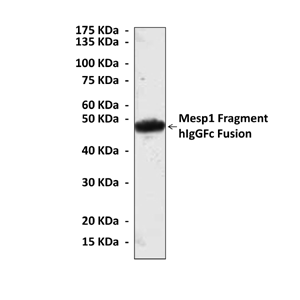
Anti-Mesp1: Mouse Mesp1 Antibody |
 |
BACKGROUND In mammals, the heart arises from the differentiation of 2 sources of multipotent cardiovascular progenitors (MCPs). Mesp1 (mesoderm posterior 1) is a key regulator of cardiovascular progenitors in vertebrates. Lineage tracing in mice demonstrated that Mesp1 represents the earliest marker of cardiovascular progenitors, tracing almost all the cells of the heart including derivatives of the primary and second heart fields. Mesp1 is a basic helix-loop-helix transcription factor that is transiently expressed by the earliest progenitors of the cardiovascular system from E6.5 to E7.5. During gastrulation, progenitors of cardiogenic mesoderm arise at E6.5 in the posterior lateral epiblast and migrate to form the cardiac crescent at E7.5, when regionalized cell fates are first delineated. Lineage tracing and heterotopic transplantation studies suggest that precursors in the earliest heart field possess potential to generate myocardium, endocardium, and epicardium, but subsequently become restricted as lineage-specific regulatory programs are activated.1 Mesp1 and Mesp2 are closely related members but share significant sequence homology only in their bHLH regions. The inactivation of Mesp1/2 indicated that Mesp genes are essential for early cardiac mesoderm formation and MCP migration. Several recent studies have demonstrated that Mesp1 massively promotes cardiovascular differentiation during embryonic development and pluripotent stem cell differentiation and indicated that Mesp1 resides at the top of the cellular and transcriptional hierarchy that orchestrates MCP specification.2 Defining how Mesp1 regulates the earliest step of MCP specification and controls their migration is essential to understand the root of cardiovascular development and how the deregulation of these processes can lead to congenital heart diseases. In addition, these findings will be very useful to boost the production of cardiovascular cells for cellular therapy, drug and toxicity screening.3
For the cellular and the molecular mechanism by which Mesp1 acts during cardiac specification, it was shown that Mesp1 acts as a cardiovascular master regulator during specification of MCPs during ESC differentiation. Embryonic stem cell (ESC) differentiation is a good model to study the cardiovascular progenitor specification. Mesp1 both directly activated many key genes belonging to the core cardiac transcriptional machinery and directly repressed genes promoting early mesoderm and endoderm cell-fate specification. Mesp1 first transiently stimulated its own endogenous expression through a direct positive autoregulatory loop and then inhibited its own expression, therefore, acting as a molecular switch during cardiac specification. Altogether, our results provide compelling evidence that Mesp1 acts as a cardiovascular master regulator during specification of MCPs during ESC differentiation.4 In addition, it was further demonstrated that Mesp1 promoted mesoderm development independently of Wnt signaling. Transient Mesp1 expression in ESCs promotes changes associated with epithelial-mesenchymal transition (EMT) and induction of Snai1, consistent with a role in gastrulation. Mesp1 expression also restricted the potential fates derived from ESCs, generating mesoderm progenitors with cardiovascular, but not hematopoietic, potential. Thus, in addition to its effects on EMT, Mesp1 may be capable of generating the multipotent cardiovascular progenitor from ESCs in vitro.5
For the cellular and the molecular mechanism by which Mesp1 acts during cardiac specification, it was shown that Mesp1 acts as a cardiovascular master regulator during specification of MCPs during ESC differentiation. Embryonic stem cell (ESC) differentiation is a good model to study the cardiovascular progenitor specification. Mesp1 both directly activated many key genes belonging to the core cardiac transcriptional machinery and directly repressed genes promoting early mesoderm and endoderm cell-fate specification. Mesp1 first transiently stimulated its own endogenous expression through a direct positive autoregulatory loop and then inhibited its own expression, therefore, acting as a molecular switch during cardiac specification. Altogether, our results provide compelling evidence that Mesp1 acts as a cardiovascular master regulator during specification of MCPs during ESC differentiation.4 In addition, it was further demonstrated that Mesp1 promoted mesoderm development independently of Wnt signaling. Transient Mesp1 expression in ESCs promotes changes associated with epithelial-mesenchymal transition (EMT) and induction of Snai1, consistent with a role in gastrulation. Mesp1 expression also restricted the potential fates derived from ESCs, generating mesoderm progenitors with cardiovascular, but not hematopoietic, potential. Thus, in addition to its effects on EMT, Mesp1 may be capable of generating the multipotent cardiovascular progenitor from ESCs in vitro.5
REFERENCES
1. Saga, Y. et al: Development 126:3437-47,1999
2. Takahashi, Y. et al: Develop. 135:787-96, 2008
3. Bondue, A. & Blanpain, C.: Circulat. Res. 107:1414-27, 2010
4. Bondue, A. et al: Cell Stem Cell 3:69-84, 2008
5. Lindsley, R.C. et al: Cell Stem Cell 3:55-68, 2008
2. Takahashi, Y. et al: Develop. 135:787-96, 2008
3. Bondue, A. & Blanpain, C.: Circulat. Res. 107:1414-27, 2010
4. Bondue, A. et al: Cell Stem Cell 3:69-84, 2008
5. Lindsley, R.C. et al: Cell Stem Cell 3:55-68, 2008
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10312 |
Antigen: | Raised against recombinant human Mesp1 proteins expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 29 kDa |
Specificity/Sensitivity: | Detects endogenous Mesp1 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой