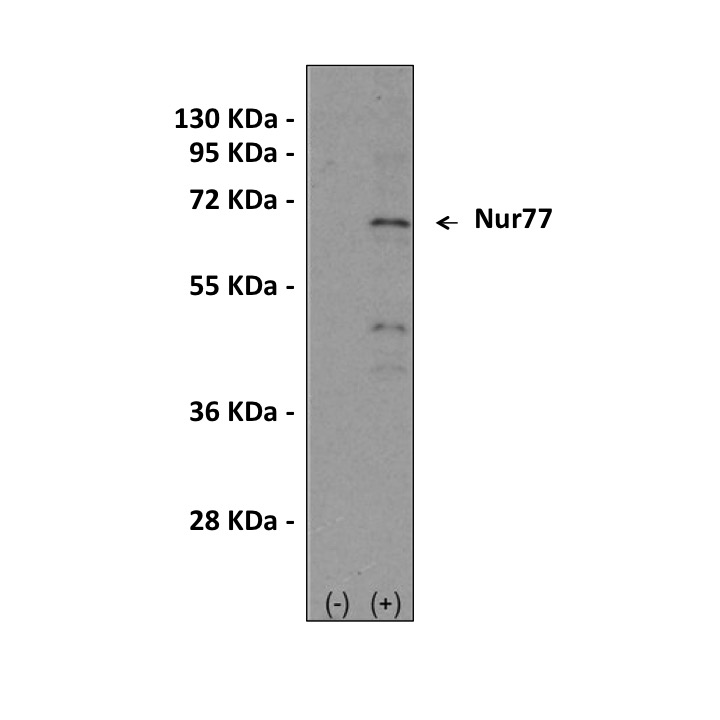
Anti-Nur77: Rabbit Nur77 Antibody |
 |
BACKGROUND The protein Nur77, also known as TR3 or Nerve Growth Factor-Induced Clone B (NGFI-B), has been implicated as a mediator of thymocyte and T cell apoptosis. Originally identified as an immediate-early gene rapidly induced by serum and growth factors including NGF, Nur77 is a member of the steroid hormone nuclear receptor superfamily and has no known ligands. It is composed of an N-terminal transactivation domain, a central-terminal DNA-binding domain, and a C-terminal ligand-binding domain.1
The Nur77 (NGFI-B) subfamily consists of three members: Nur77 (NGFI-Balpha), Nurr1 (NGFI-Bbeta), and Nor1 (NGFI-Bgamma). The DNA-binding domains of these proteins are >90% homologous, whereas the ligand-binding domains are intermediately conserved in this subfamily. In contrast, the sequences of the transactivation domain are not well conserved. Although Nur77 is widely expressed in various tissues, Nurr1 is detected primarily in thymus, osteoblasts, liver, and pituitary gland, and Nor1 is expressed at a high level in the pituitary gland but at low levels in thymus, kidney, heart, skeletal muscle, and adrenal glands. Nur77 subfamily members have been shown to bind DNA as monomers, homodimers, or heterodimers with retinoid X receptor. Nur77 is constitutively active when overexpressed, suggesting the possibility that Nur77 activity does not require ligand stimulation.
Along with the two other Nur family members, Nur77 is expressed in macrophages following inflammatory stimuli. This process is mediated by the NF-κB (nuclear factor-kappa B) complex, a ubiquitous transcription factor involved in cellular response to stress. It can also be induced by many physiological and physical stimuli. These include physiological stimuli such as "fatty acids, stress, prostaglandins, growth factors, calcium, inflammatory cytokines, peptide hormones, phorbol esters, and neurotransmitters" and physical stimuli including "magnetic fields, mechanical agitation (causing fluid shear stress), and membrane depolarization.2 In addition, It has been reported that Nur77 is inducible by T cell receptor signals and that its expression is high during T cell receptor-mediated apoptosis. Inhibition of Nur77 function by a dominant-negative (DN) Nur77 blocks T cell receptor-mediated apoptosis in thymocytes as well as in the negative selection, whereas thymocytes from Nur77-expressing transgenic mice show massive apoptosis. However, Nur77 knockout mice do not exhibit any defect in thymocyte apoptosis, suggesting a potential functional redundancy among Nur77 family members. Although it remains unclear how Nur77 functions to mediate apoptosis, it was suggested that, in response to certain apoptotic stimuli, Nur77 is translocated from the nucleus to the mitochondria, where it induces cytochrome c release. In this scenario, the transactivation activity of Nur77 may not be required for its proapoptotic effect. However, it has also been shown that Akt/PKB can phosphorylate Nur77 at Ser351, which inhibits the functions of Nur77 as a transcription factor and as a mediator of apoptosis in T cells.3 In addition, it was shown that that Nur77 is a survival effector protein in the context of TNF-mediated signaling.4
REFERENCES
1. Hsu, H. et al: Curr. Drug Target-Inflamm. Allerg. 3:413-23, 2004
2. Pei, L. et al: Mol. Endocrinol. 20:786-94, 2006
3. Masuyama, N. et al: J. Biol. Chem. 276:32799-805, 2001
4. Suzuki, S. Et al: Proc. Natl. Acad. Sci. USA 100:8276-80, 2003
2. Pei, L. et al: Mol. Endocrinol. 20:786-94, 2006
3. Masuyama, N. et al: J. Biol. Chem. 276:32799-805, 2001
4. Suzuki, S. Et al: Proc. Natl. Acad. Sci. USA 100:8276-80, 2003
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CG1365 |
Antigen: | A short peptide from N-terminal sequence of human Nur77. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 64463 Da |
Specificity/Sensitivity: | Detects endogenous Nur77 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой