Anti-SLC22A1/OCT1: Mouse SLC22A1/OCT1 Antibody |
 |
BACKGROUND Transporter proteins have a major role in the absorption, distribution, and elimination of a wide variety of compounds. Organic cation transporters (OCT) are involved in the carriage of both endogenous compounds, such as dopamine, and drugs, such as metformin and some anti-Parkinsonian drugs. The three most important members of the OCT family are OCT1, OCT2, and OCT3, encoded by the SLC22A1, SLC22A2, and SLC22A3 genes, respectively. They are generally present in the liver, kidney, and intestine. The polyspecific organic cation transporters OCT1, 2, and 3 mediate the facilitated transport of a variety of structurally diverse organic cations. OCT1 and OCT2 are found in the basolateral membrane of hepatocytes, enterocytes, and renal proximal tubular cells. OCT3 has a more widespread tissue distribution and is considered to be the major component of the extraneuronal monoamine transport system (or uptake-2), which is responsible for the peripheral elimination of monoamine neurotransmitters. Studies with knockout mouse models have directly demonstrated that these transporters can have a major impact on the pharmacological behavior of various substrate organic cations.1 The recent identification of polymorphic genetic variants of human OCT1 and OCT2 that severely affect transport activity thus suggests that some of the interpatient differences in response and sensitivity to cationic drugs may be caused by variable activity of these transporters.2
SLC22A1/OCT1 is highly expressed in both human and rodent liver and is the major organic cationic transport protein responsible for hepatocellular organic cationic drug uptake. Therefore, SLC22A1/OCT1-mediated uptake into the liver is an essential step in the biotransformation of many pharmacological substances and nutrients. In vivo studies using Oct1-/- mice and clinical pharmacokinetic/ pharmacogenetic analyses have demonstrated that hepatic OCT1 is responsible for the therapeutic efficacy and toxicity of a biguanide agent, metformin. In addition, SLC22A1/OCT1 transports the organic cation choline, which is an essential step in the synthesis of phosphatidylcholine via the Kennedy pathway. It was shown that PPAR-alpha and –gamma agonists transcriptionally increase SLC22A1/OCT1 gene expression, and the increased SLC22A1/OCT1 expression results in enhanced cellular organic cation uptake. Thus, there may be an implication for the uptake of organic cationic drugs and for lipid metabolism.3
SLC22A1/OCT1 is highly expressed in both human and rodent liver and is the major organic cationic transport protein responsible for hepatocellular organic cationic drug uptake. Therefore, SLC22A1/OCT1-mediated uptake into the liver is an essential step in the biotransformation of many pharmacological substances and nutrients. In vivo studies using Oct1-/- mice and clinical pharmacokinetic/ pharmacogenetic analyses have demonstrated that hepatic OCT1 is responsible for the therapeutic efficacy and toxicity of a biguanide agent, metformin. In addition, SLC22A1/OCT1 transports the organic cation choline, which is an essential step in the synthesis of phosphatidylcholine via the Kennedy pathway. It was shown that PPAR-alpha and –gamma agonists transcriptionally increase SLC22A1/OCT1 gene expression, and the increased SLC22A1/OCT1 expression results in enhanced cellular organic cation uptake. Thus, there may be an implication for the uptake of organic cationic drugs and for lipid metabolism.3
REFERENCES
1. Nies, A.T. et al: Drug Transport.201:105-67, 2011
2. Itoda, M. et al: Drug Metab. and Pharmacokin. 19:308-12, 2004
3. Nie, W. et al: Am. J. Physiol. Gastrointest. Liver Physiol. 288: G207–G212, 2005
2. Itoda, M. et al: Drug Metab. and Pharmacokin. 19:308-12, 2004
3. Nie, W. et al: Am. J. Physiol. Gastrointest. Liver Physiol. 288: G207–G212, 2005
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10360 |
Antigen: | Raised against recombinant human SLC22A1/OCT1 fragments expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC n/d FACS 1:50 - 1:200 |
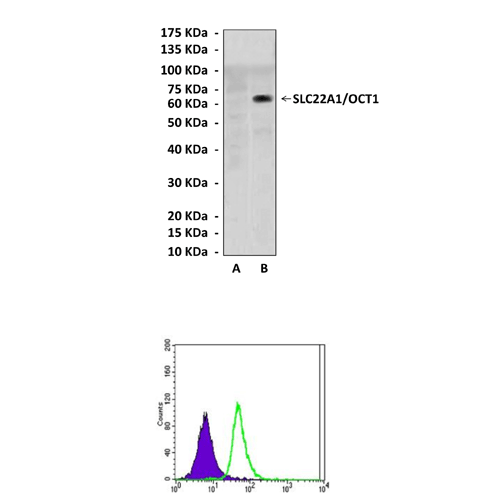
Predicted Molecular Weight of protein: | 62 kDa |
Specificity/Sensitivity: | Detects SLC22A1/OCT1 proteins in various cell lysate. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой