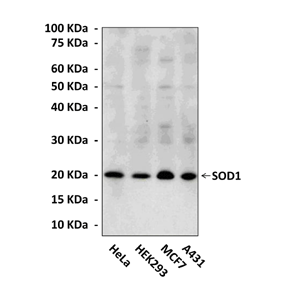
Anti-SOD1: Monoclonal Superoxide Dismutase 1 Antibody (48C10) |
 |
BACKGROUND Superoxide dismutase 1 (SOD1) is a human superoxide dismutase. In humans (as in all other mammals and most chordates), three forms of superoxide dismutase are present. SOD1 is located in the cytoplasm, SOD2 in the mitochondria and SOD3 is extracellular. The first is a dimer (consists of two units), while the others are tetramers (four subunits). SOD1 and SOD3 contain copper and zinc, while SOD2 has manganese in its reactive centre.1 SOD1 and others are responsible for destroying free superoxide radicals in the body by converting naturally occurring, but harmful, superoxide radicals to molecular oxygen and hydrogen peroxide. Mutations (over 100 identified to date) in this gene cause familial amyotrophic lateral sclerosis.2 The most frequent mutation are A4V (in the U.S.A.) and H46R (Japan). The most studied ALS mouse model is G93A. Rare transcript variants have been reported for this gene. Mice lacking Sod1 have increased age-related muscle mass loss (sarcopenia), early development of cataracts, macular degeneration, thymic involution, hepatocellular carcinoma, and shortned lifespan.3, 4
REFERENCES
1. Zelko, I.N. et al: Free Radic. Biol. Med. 33:337, 2003.
2. de Belleroche, J. et al: J. Med. Genet. 32:841, 1996.
3. Elchuri, S. et al: Oncogene 24:367, 2005.
4. Sentman, M. L.et al: J. Biol. Chem. 281:6904, 2006.
2. de Belleroche, J. et al: J. Med. Genet. 32:841, 1996.
3. Elchuri, S. et al: Oncogene 24:367, 2005.
4. Sentman, M. L.et al: J. Biol. Chem. 281:6904, 2006.
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CB14379 |
Antigen: | E. coli-expressed recombinant human SOD1 protein fragments. |
Isotype: | Mouse Monoclonal IgG1 |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions: | WB 1:1000 IP 1:50 IHC (Paraffin) 1:100 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 18 kDa |
Specificity/Sensitivity: | Specifically detects endogenous levels of human SOD1 proteins. Does not cross-react with other related proteins. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой