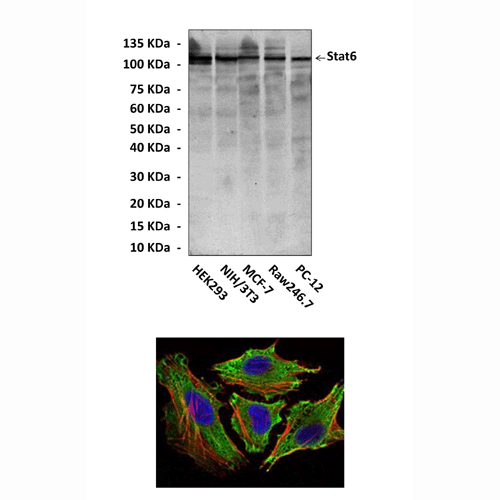
Anti-Stat6: Mouse Stat6 Antibody |
 |
BACKGROUND Signal Transducers and Activators of Transcription (STATs) were first discovered in protein complexes bound to the promoters of interferon-inducible genes. Seven STATs have now been cloned: STAT1, STAT2, STAT3, STAT4, STAT5A, STAT5B, and STAT6. These transcription factors participate in normal cellular events, such as differentiation, proliferation, cell survival, apoptosis, and angiogenesis following cytokine, growth factor, and hormone signaling.1 STATs are activated by tyrosine phosphorylation, which is normally a transient and tightly regulates process. The seven mammalian STATs bear a conserved tyrosine residue near the C-terminus that is phosphorylated by JAKs. This phosphotyrosine permits the dimerization of STATs through interaction with a conserved SH2 domain. Phosphorylated STATs enter the nucleus by a mechanism that is dependent on importin-5 (also called nucleoprotein interactor 1) and the Ran nuclear import pathway. Once in the nucleus, dimerized STATs bind specific regulatory sequences to activate or repress transcription of target genes. Thus the JAK/STAT cascade provides a direct mechanism to translate an extracellular signal into a transcriptional response.2 In addition, serine phosphorylation of STATs may be required for the maximal induction of Stat-mediated gene activation.3 STATs can be divided into two groups according to their specific functions. One is made up of STAT2, STAT4, and STAT6, which are activated by a small number of cytokines and play a distinct role in the development of T-cells and in IFN signaling. The other group includes STAT1, STAT3, and STAT5, activated in different tissues by means of a series of ligands and involved in IFN signaling, development of the mammary gland, response to GH, and embriogenesis. This latter group of STATS plays an important role in controlling cell-cycle progression and apoptosis and thus contributes to oncogenesis.
Stat1 protein exists as a pair of isoforms, Stat1α (91 kDa) and the splice variant Stat1-beta (84 kDa). Although an increased expression of STAT1 has been observed in many human neoplasias, this molecule can be considered a potential tumor suppressor, since it plays an important role in growth arrest and in promoting apoptosis.4 On the other hand, STAT3 and 5 are considered as oncogenes, since they bring about the activation of cyclin D1, c-Myc, and bcl-xl expression, and are involved in promoting cell-cycle progression, cellular transformation, and in preventing apoptosis.5
Stat6 plays a central role in exerting IL4 mediated biological responses. STAT6 mediates many of the functions of IL-4 downstream of the IL-4R. Importantly, mice that lack IL-4R-alpha or STAT6 are highly resistant to experimentally induced allergic airway inflammation and show significant defects in Th2 differentiation. Additionally, Stat6 is found to induce the expression of BCL2L1/BCL-X(L), which is responsible for the anti-apoptotic activity of IL4. Knockout studies in mice suggested the roles of Stat6 in differentiation of T helper 2 (Th2) cells, expression of cell surface markers, and class switch of immunoglobulins.6
Stat1 protein exists as a pair of isoforms, Stat1α (91 kDa) and the splice variant Stat1-beta (84 kDa). Although an increased expression of STAT1 has been observed in many human neoplasias, this molecule can be considered a potential tumor suppressor, since it plays an important role in growth arrest and in promoting apoptosis.4 On the other hand, STAT3 and 5 are considered as oncogenes, since they bring about the activation of cyclin D1, c-Myc, and bcl-xl expression, and are involved in promoting cell-cycle progression, cellular transformation, and in preventing apoptosis.5
Stat6 plays a central role in exerting IL4 mediated biological responses. STAT6 mediates many of the functions of IL-4 downstream of the IL-4R. Importantly, mice that lack IL-4R-alpha or STAT6 are highly resistant to experimentally induced allergic airway inflammation and show significant defects in Th2 differentiation. Additionally, Stat6 is found to induce the expression of BCL2L1/BCL-X(L), which is responsible for the anti-apoptotic activity of IL4. Knockout studies in mice suggested the roles of Stat6 in differentiation of T helper 2 (Th2) cells, expression of cell surface markers, and class switch of immunoglobulins.6
REFERENCES
1. Artavanis-Tsakonas S et al.: Science 284: 770-776, 1999.
2. Oswald F et al.: Mol. Cell. Biol. 21: 7761–74, 2001.
3. Laky K & Fowlkes BJ: Curr. Opin. Immunol. 20: 197-202, 2008.
4. Chen, S. & Schreiber, S.:Cancer Res. 69(24 Suppl):Abstract nr 65, 2009
5. Calò, V. et al: J. Cell. Physiol. 197:157-68, 2003
6. Sherman, M.A.:Immunol. Rev.179:48-56, 2001
2. Oswald F et al.: Mol. Cell. Biol. 21: 7761–74, 2001.
3. Laky K & Fowlkes BJ: Curr. Opin. Immunol. 20: 197-202, 2008.
4. Chen, S. & Schreiber, S.:Cancer Res. 69(24 Suppl):Abstract nr 65, 2009
5. Calò, V. et al: J. Cell. Physiol. 197:157-68, 2003
6. Sherman, M.A.:Immunol. Rev.179:48-56, 2001
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10329 |
Antigen: | Raised against recombinant human Stat6 fragments expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP 1:50 IHC n/d ICC 1:50 - 1:200 FACS n/d |
Predicted Molecular Weight of protein: | 105 kDa |
Specificity/Sensitivity: | Detects endogenous Stat6 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой