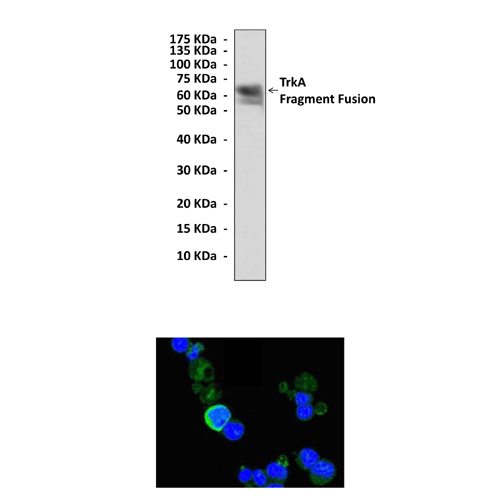
Anti-TrkA: Mouse Neurotrophin A Antibody |
 |
BACKGROUND Trk (neurotrophin) receptors are single transmembrane catalytic receptors with intracellular tyrosine kinase activity. Trk receptors are coupled to the Ras, Cdc42/Rac/RhoG, MAPK, PI 3-K and PLCgamma signaling pathways. There are four members of the Trk family; TrkA, TrkB and TrkC and a related p75NTR receptor. p75NTR lacks tyrosine kinase activity and signals via NF-kappaB activation. Each family member binds different neurotrophins with varying affinities. TrkA potently binds Nerve Growth Factor (NGF) and is involved in differentiation and survival of neurons and in control of gene expression of enzymes involved in neurotransmitter synthesis. TrkB has highest affinity for brain-derived neurotrophic factor (BDNF) and is involved in neuronal plasticity, long term potentiation and apoptosis of CNS neurons. TrkC is activated by neurotrophin-3 (NT-3) and is found on proprioceptive sensory neurons. p75NTR binds neurotrophin precursors with high affinity and retains low affinity to the mature cleaved forms. TrkA was originally identified as an oncogene as it is commonly mutated in cancers, particularly colon and thyroid carcinomas.1
TrkA is typically activated after engagement with the neurotrophins nerve growth factor (NGF) during development and the repair of the nervous system. Mutations in TrkA gene have been associated with congenital insensitivity to pain, anhidrosis, self-mutilating behavior, mental retardation and cancer. Three alternate transcriptional splice variants of TrkA gene have been found. Activation of TrkA-I (neuronal) or TrkA-II (nonneuronal) isoforms by NGF leads to TrkA activation and signaling, ultimately resulting in survival and neuronal differentiation. In the absence of ligand, alternative pathways are activated leading to apoptosis. The TrkA-III isoform is constitutively active, ligand independent, and promotes survival mainly through the PI3K-AKT pathway. It may be also a novel internal membrane-associated centrosome kinase and involved in oncogenesis process.2
The TrkA signal transduction involves several pathways. Ligand binding causes receptor homodimerization, which activates its tyrosine kinase activity and leads to transphosphorylation on at least five tyrosine residues. Autophosphorylation provides docking sites for downstream effectors through SH2 domains including SHC, PI3K, and PLCγ1, which are the primary effectors of Trk activity in NGF-treated PC12 cells. Stat3 has been recently identified as a novel signal transducer for TrkA. It was shown that that TrkA oncogenes trigger STAT3 phosphorylation both on Y705 and S727 residues and STAT3 transcriptional activity.3 Other important signaling proteins include SH2B/APS, FRS2, FRS3, IRS1, IRS2, Akt and SHP-1. SH2B and FRS2 activation are important for neuronal differentiation, whereas AKT activation is important for survival. Additionally, it was also demonstrated that NGF signaling via the trkA receptor requires formation of a signaling endosome containing NGF and trkA. Thus, endocytosis of the neurotrophin–receptor complex is a crucial step in the generation of intracellular signaling platforms required for activation and compartmentalization of signaling events.4 Furthermore, activation of TrkA receptors also may have different consequences, depending on the cellular context. For example, exposure of rat PC12 pheochromocytoma cells to NGF causes neuronal differentiation, but NGF exposure of mouse NIH-3T3 fibroblasts transfected with TrkA leads to enhanced cell proliferation.5
TrkA is typically activated after engagement with the neurotrophins nerve growth factor (NGF) during development and the repair of the nervous system. Mutations in TrkA gene have been associated with congenital insensitivity to pain, anhidrosis, self-mutilating behavior, mental retardation and cancer. Three alternate transcriptional splice variants of TrkA gene have been found. Activation of TrkA-I (neuronal) or TrkA-II (nonneuronal) isoforms by NGF leads to TrkA activation and signaling, ultimately resulting in survival and neuronal differentiation. In the absence of ligand, alternative pathways are activated leading to apoptosis. The TrkA-III isoform is constitutively active, ligand independent, and promotes survival mainly through the PI3K-AKT pathway. It may be also a novel internal membrane-associated centrosome kinase and involved in oncogenesis process.2
The TrkA signal transduction involves several pathways. Ligand binding causes receptor homodimerization, which activates its tyrosine kinase activity and leads to transphosphorylation on at least five tyrosine residues. Autophosphorylation provides docking sites for downstream effectors through SH2 domains including SHC, PI3K, and PLCγ1, which are the primary effectors of Trk activity in NGF-treated PC12 cells. Stat3 has been recently identified as a novel signal transducer for TrkA. It was shown that that TrkA oncogenes trigger STAT3 phosphorylation both on Y705 and S727 residues and STAT3 transcriptional activity.3 Other important signaling proteins include SH2B/APS, FRS2, FRS3, IRS1, IRS2, Akt and SHP-1. SH2B and FRS2 activation are important for neuronal differentiation, whereas AKT activation is important for survival. Additionally, it was also demonstrated that NGF signaling via the trkA receptor requires formation of a signaling endosome containing NGF and trkA. Thus, endocytosis of the neurotrophin–receptor complex is a crucial step in the generation of intracellular signaling platforms required for activation and compartmentalization of signaling events.4 Furthermore, activation of TrkA receptors also may have different consequences, depending on the cellular context. For example, exposure of rat PC12 pheochromocytoma cells to NGF causes neuronal differentiation, but NGF exposure of mouse NIH-3T3 fibroblasts transfected with TrkA leads to enhanced cell proliferation.5
REFERENCES
1. Lamballe, F. et al: Cell 66:967-979, 1991
2. Farina, A.R. et al: Mol. Cell Biol. 29:4812-30, 2009
3. Miranda, C. et al: PloS ONE 5:e9446, 2010
4. Barker, P.A. et al: Trends Neurosci. 25:379-38, 2002
5. Brodeur, G.M. et al: Clin Cancer Res. 15:3244-50, 2009
2. Farina, A.R. et al: Mol. Cell Biol. 29:4812-30, 2009
3. Miranda, C. et al: PloS ONE 5:e9446, 2010
4. Barker, P.A. et al: Trends Neurosci. 25:379-38, 2002
5. Brodeur, G.M. et al: Clin Cancer Res. 15:3244-50, 2009
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10239 |
Antigen: | Purified recombinant human TrkA extracellular domain-hIgG-Fc fusion proteins expressed in 293 cells. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000 IP 1:50 IHC n/d ICC 1:100 - 1:200 FACS 1:50 |
Predicted Molecular Weight of protein: | 140 kDa |
Specificity/Sensitivity: | Detects endogenous TrkA proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой