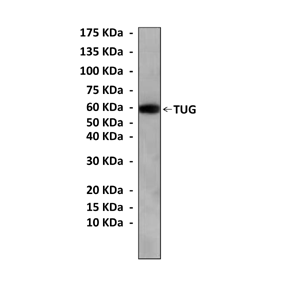
Anti-TUG: Mouse TUG Antibody |
 |
BACKGROUND Insulin stimulates glucose uptake in fat and muscle by redistributing GLUT4 glucose transporters from intracellular membranes to the cell surface. In unstimulated cells, GLUT4 is retained in intracellular membranes, and its predominant exclusion from the plasma membrane minimizes glucose uptake from the extracellular space. Within minutes of insulin stimulation, GLUT4 is translocated so that the number of transporters in the plasma membrane is increased and glucose uptake is accelerated. This insulin action is defective in muscle and adipose from individuals with type 2 diabetes and other insulin-resistant states. To redistribute GLUT4, insulin acts primarily on the exocytic arm of the GLUT4 recycling pathway. The signaling proteins involved include Akt/protein kinase B and its substrate AS160; the latter protein was recently shown also to contribute to intracellular retention of GLUT4 in unstimulated cells. Studies of GLUT4 vesicle traffic have been complicated by the fact that unstimulated cells contain multiple pools of intracellular GLUT4. These include GLUT4 in endosomes, the trans-Golgi network, and poorly characterized membranes sometimes termed as the “insulin-responsive compartment” or “GLUT4 storage vesicles” (GSVs). Insulin stimulates endosome recycling, which may contribute a 2-fold increase in cell surface GLUT4 and other proteins such as the transferrin receptor (TfnR). The more dramatic, insulin-stimulated increase in cell surface GLUT4, and not in other recycling proteins, is due to mobilization of GSVs. These membranes comprise small, preformed vesicles that are depleted of TfnR and other endosomal markers. Data indicate that GSVs are derived by budding from the trans-Golgi network, yet they may also exchange with endosomes. In adipocytes, GSVs are distinct from secretory vesicles containing adiponectin. The biogenesis of these vesicles is both cell type-specific and developmentally regulated.1
In unstimulated cells, GLUT4 that is present in GSVs may be tethered to intracellular structures to restrict its movement to the plasma membrane. Insulin “untethers” this GLUT4 to accelerate its movement to the cell surface and subsequent fusion at the plasma membrane. TUG (tether, containing a UBX domain, for GLUT4) is a putative tethering protein, which acts in concert with other proteins to retain GLUT4 within cells in the absence of insulin.2 As a cytosolic protein, TUG binds directly and specifically to GLUT4 and not GLUT1. TUG and GLUT4 form a complex in unstimulated 3T3-L1 adipocytes, and colocalize on TfnR-negative intracellular membranes that sediment as light microsomes. These are properties shared by GSVs. The TUG-GLUT4 complex is insulin-responsive, because insulin acts rapidly to stimulate the dissociation of TUG and GLUT4 and releasing these vesicles to the cellular exocytic machinery for translocation to the plasma membrane. Overexpression of TUG increases the size of the insulin-responsive GLUT4 pool, assessed using kinetics, and increases the number of insulin-responsive TUG-GLUT4 protein complexes. Conversely, expression of a dominant negative TUG UBX-Cter fragment decreases the size of the insulin-responsive pool and decreases the number of TUG-GLUT4 complexes. Thus, TUG is part of an insulin-responsive mechanism governing the sequestration of GLUT4 in GSVs within unstimulated 3T3-L1 cells. TUG is required to retain GLUT4 intracellularly in 3T3-L1 adipocytes in the absence of insulin and the insulin-stimulated dissociation of TUG and GLUT4 is an important mechanism by which insulin stimulates glucose uptake.3 TUG is also found to regulate skeletal muscle Glut4 traffic in transgenic mouse model.4
In unstimulated cells, GLUT4 that is present in GSVs may be tethered to intracellular structures to restrict its movement to the plasma membrane. Insulin “untethers” this GLUT4 to accelerate its movement to the cell surface and subsequent fusion at the plasma membrane. TUG (tether, containing a UBX domain, for GLUT4) is a putative tethering protein, which acts in concert with other proteins to retain GLUT4 within cells in the absence of insulin.2 As a cytosolic protein, TUG binds directly and specifically to GLUT4 and not GLUT1. TUG and GLUT4 form a complex in unstimulated 3T3-L1 adipocytes, and colocalize on TfnR-negative intracellular membranes that sediment as light microsomes. These are properties shared by GSVs. The TUG-GLUT4 complex is insulin-responsive, because insulin acts rapidly to stimulate the dissociation of TUG and GLUT4 and releasing these vesicles to the cellular exocytic machinery for translocation to the plasma membrane. Overexpression of TUG increases the size of the insulin-responsive GLUT4 pool, assessed using kinetics, and increases the number of insulin-responsive TUG-GLUT4 protein complexes. Conversely, expression of a dominant negative TUG UBX-Cter fragment decreases the size of the insulin-responsive pool and decreases the number of TUG-GLUT4 complexes. Thus, TUG is part of an insulin-responsive mechanism governing the sequestration of GLUT4 in GSVs within unstimulated 3T3-L1 cells. TUG is required to retain GLUT4 intracellularly in 3T3-L1 adipocytes in the absence of insulin and the insulin-stimulated dissociation of TUG and GLUT4 is an important mechanism by which insulin stimulates glucose uptake.3 TUG is also found to regulate skeletal muscle Glut4 traffic in transgenic mouse model.4
REFERENCES
1. Bryant, N.J. et al: Nat. Rev. Mol. Cell. Biol. 3:267-77, 2002
2. Bogan, J.S. et al: Nature 425:7727-33, 2003
3. Yu, C. et al: J. Biol. Chem. 282:7710-22, 2007
4. Schertzer, J. D. et al: Endocrinol. 150:1935-40, 2009
2. Bogan, J.S. et al: Nature 425:7727-33, 2003
3. Yu, C. et al: J. Biol. Chem. 282:7710-22, 2007
4. Schertzer, J. D. et al: Endocrinol. 150:1935-40, 2009
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10241 |
Antigen: | Purified recombinant human TUG fragments expressed in E. coli. |
Isotype: | Mouse IgG |
Species & predicted species cross- reactivity ( ): | Human, Mouse |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 60 kDa |
Specificity/Sensitivity: | Detects endogenous TUG proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой